-
Články
- Časopisy
- Kurzy
- Témy
- Kongresy
- Videa
- Podcasty
- Kariéra
An unusual case of late recurrent adult granulosa cell tumor and mature teratoma arising within the same ovary, confirmed by NGS analysis
Neobvyklý a molekulárně-biologicky verifikovaný případ recidivujícího adultního nádoru z buněk granulózy vznikající společně se zralým teratomem ve stejném ovariu
Adultní nádor z buněk granulózy je nejčastějším maligním gonadostromálním nádorem ovaria, představuje přibližně 3–5 % všech malignit ovaria a je známý svým rizikem recidiv spojených s vysokou mortalitou. Představujeme zajímavou kazuistiku 71leté ženy s doposud prvním zdokumentovaným případem recidivujícího adultního nádoru z buněk granulózy vznikajícího společně se zralým teratomem ve stejném ovariu, potvrzeným jak imunohistochemií, tak molekulárně-biologickou analýzou. Nádor vykazoval v primární i recidivující lézi missense mutace v genu FOXL2, která je typická pro adultní nádor z buněk granulózy. Mutace v genu TP53, TSC2 a RB1 byly přítomny pouze v recidivujícím nádoru, což naznačuje sekundární mutace získané během progrese.
Klíčová slova:
teratóm – ovarium – NGS – sex cord-stromální nádory – adultní nádor z buněk granulózy
Authors: Adam Šafanda; Nikola Hájková; Jan Galko; Michaela Kendall Bártů; Pavel Dundr; Kristýna Němejcová
Authors place of work: Department of Pathology, First Faculty of Medicine, Charles University and General University Hospital in Prague, Prague, Czech Republic
Published in the journal: Čes.-slov. Patol., 61, 2025, No. 4, p. 206-209
Category: Původní práce
Summary
Adult granulosa cell tumor is a predominant malignant tumor among ovarian sex cord-stromal tumors, representing approximately 3-5% of all ovarian malignancies and being known for its risk of recurrence with high mortality rate. We present a unique case of a 71-year-old woman with, to our knowledge, the first documented instance of a recurrent AGCT arising concurrently with a mature ovarian teratoma, confirmed through both immunohistochemistry and molecular biological analysis. The tumor in both the primary and recurrent lesion harbored a missense FOXL2 mutation typical for adult granulosa cell tumor. TP53, TSC2 and RB1 mutations were present only in the recurrent tumor, indicating secondary mutations acquired during progression.
Keywords:
teratoma – ovary – NGS – sex cord-stromal tumors – adult granulosa cell tumor of the ovary
* Correspondence address:
Doc. MUDr. Kristýna Němejcová, Ph.D
Department of Pathology
First Faculty of Medicine, Charles University and General University Hospital in Prague
Studničkova 2, 12800 Prague 2, Czech Republic
tel.: +420224968632
e-mail: kristyna.nemejcova@vfn.czOvarian sex cord-stromal tumors comprise a heterogeneous group of neoplasms characterized by variable biological behavior, classified into three categories according to the cellular origin; including pure stromal tumors, pure sex cord tumors, and mixed sex cord-stromal tumors (1). Adult granulosa cell tumor (AGCT) is the most prevalent malignant subtype within the sex cord-stromal tumor group, accounting for approximately 3% to 5% of all ovarian malignancies. A subset of ovarian sex cord-stromal tumors exhibits uncertain biological behavior, with AGCT in particular prone to late recurrences which significantly reduce the survival rate (2-3). Recent advancements in molecular biology have facilitated the resolution of challenging cases where morphological and immunohistochemical features overlap, for example in case of missense FOXL2 (c.402C>G, p.Cys134Trp) mutation which is mostly found in AGCT and a subset of Sertoli Leydig cell tumors (SLCT), as described in our recent publication (4-6). Mature teratomas are a type of germ cell tumor and represent one of the most common ovarian neoplasms. They are characterised by the presence of differentiated tissues derived from all three germ layers: ectoderm, mesoderm, and endoderm (1,7). Mature teratomas are benign tumors, in contrast to immature teratomas, which are malignant and are graded based on the amount of immature neuroectodermal tissue (1). However, malignant transformation of mature teratoma occurs in approximately 2% of cases with squamous cell carcinoma being the most common type of somatic malignancy arising in a mature teratoma (8). Various other somatic malignancies have been documented arising within teratomas, including rare cases of sex cord-stromal tumors, exclusively AGCT (7,9). Nevertheless, most of the existing literature describes AGCT and mature teratoma as independent synchronous neoplasms, in contrast to secondary somatic malignancies arising within a pre-existing mature teratoma (7). We present a unique case of a 71-year-old woman with, to our knowledge, the first documented instance of a recurrent AGCT arising concurrently with a mature ovarian teratoma, confirmed through both immunohistochemistry and molecular biological analysis.
CLINICAL HISTORY
The patient was a 71-year-old female with diffuse large B-cell lymphoma (DLBCL) in personal history, who underwent bilateral adnexectomy and hysterectomy in 2007 with an incidental finding of a mature cystic teratoma of the left ovary. In 2023 imaging revealed a sigmoid colon mass and multiple liver lesions suspicious for metastases, which were treated by resection of the sigmoid colon and a biopsy of the liver lesions, followed by subsequent chemotherapy. As of November 2024, the patient remains alive with active disease and is currently undergoing chemotherapy to manage the liver metastases.
MATERIALS AND METHODS
Immunohistochemical analysis
Immunohistochemical (IHC) analysis was performed using 4-μm-thick sections of formalin-fixed and paraffin-embedded (FFPE) tissue. The list of antibodies used, their clones, manufacturers, dilution, and staining instruments is summarized in Table 1.
Molecular analysis
Genomic DNA was isolated from FFPE tissue from the recurrent AGCT (estimated tumor purity was 95 % of tumor cells) and from the AGCT associated with a mature teratoma of the ovary (80 % of tumor cells) using the Quick-DNA/RNA FFPE Miniprep Kit (Zymo Research), according to the manufacturer’s protocol.
Sequence capture NGS analysis of the DNA was performed using the KAPA HyperPlus kit according to KAPA HyperCap Workflow v3.0 (Roche) and a panel of hybridization probes against multiple targets of cancer relevant genes (360 genes or gene parts; 951 kbp of coding regions; Roche). Unique Molecular Identifiers (UMIs) were incorporated during library preparation in the primary tumor (from 2007) to remove sequencing errors, unmasking low-frequency variants. The prepared sample libraries were pair-end sequenced by the NextSeq 500 instrument (Illumina) using NextSeq 500/550 High Output Kit v2.5 (Illumina). The biostatistical evaluation was performed using CLC Genomics Workbench software (CLC GW; Qiagen, Venlo, The Netherlands) and variants with mutation allele frequency (MAF) of at least 4 % were identified. Interpretation of the DNA variants was performed as described in a previous study (10). All variants were manually reviewed using the IGV software to eliminate potential sequencing artifacts or PCR duplicates and were analyzed and compared between both tumors. A variant with ≥ 1% MAF detected in the primary tumor was considered present if it was also detected in the recurrent tumor.
RESULTS
Morphological and immunohistochemical findings
The sigmoid colon mass measured 80x70x60 mm, presenting as a homogenous yellow mass with focal areas of necrosis, and was situated within the muscularis externa and subserosal layers of the colon. Histologically, the tumor exhibited features of AGCT with predominantly diffuse growth pattern, with areas of trabecular and alveolar structures and multiple necrotic areas. The tumor cells displayed eosinophilic cytoplasm and relatively uniform, larger vesicular nuclei with prominent nucleoli. In certain regions the tumor showed marked nuclear atypia, including the presence of monstrous nuclei and frequent mitotic figures. During the diagnostic evaluation of the sigmoid colon tumor, the left ovarian tumor diagnosed in 2007 as mature teratoma was re-examined, and the second reading showed focal areas of a tumor with AGCT features growing in a close proximity to the mature teratoma, which was made up of structures of a dermoid cyst, thyroid gland tissue, and mucinous epithelium. The biopsy of one of the liver lesions showed a tumor of the same morphology of AGCT as the mass in the sigmoid colon. Representative examples of the microscopic features (H&E staining) of the primary and recurrent tumors, along with the results of SF1 and p53 immunohistochemistry of the recurrent tumor are presented in Figure 1. The results of the immunohistochemical examination are provided in Table 1.
Table 1. List of the immunohistochemical antibodies used, including their clones, manufacturers, dilution, staining instruments, and a summary of the results in the primary and recurrent adult granulosa cell tumor.
Antibody
Clone
Dilution
Producer
Platform
Detection
Results (primary tumor)
Results (recurrence)
SF1
EPR 19744
1 : 400
Abcam, Cambridge, UK
Dako Omnis, (Agilent, Santa Clara, CA, USA
EnVision FLEX (Dako)
Diffuse and strong expression
Diffuse and strong expression
Calretinin
DAKCalret
1 : 00
Dako, Glostrup, Denmark
Dako Omnis, (Agilent, Santa Clara, CA, USA
EnVision FLEX (Dako)
Diffuse and strong expression
Moderate expression in 5% of cells
Inhibin A
R1
RTU
Dako, Glostrup, Denmark
Dako Omnis, (Agilent, Santa Clara, CA, USA
EnVision FLEX (Dako)
Diffuse and strong expression
Moderate expression in 5-10% of cells
FOXL2
Polyclonal Ra
1 : 200
Novus Bio, USA
Ventana BenchMark ULTRA (Roche, Basel, Switzerland)
OptiView
Weak to moderate extensive (80%) expression
Weak to moderate expression in 60% of cells
CKAE 1/3
AE1/AE3
1 : 200
Dako, Glostrup, Denmark
Dako Omnis, (Agilent, Santa Clara, CA, USA
EnVision FLEX (Dako)
Granular positivity in 50% of cells
Moderate expression in 30-40% of cells
p53
DO-7
1 : 400
Dako, Glostrup, Denmark
Ventana BenchMark ULTRA (Roche, Basel, Switzerland)
UltraView
Wild type
Aberrant
ER
SP1
1 : 200
Zytomed Systems GmbH, Berlin, Germany
Ventana BenchMark ULTRA (Roche, Basel, Switzerland)
OptiView
Weak expression in 1020% of cells
Weak expression in 1020% of cells
MAF: mutation allele frequency; AGCT: adult granulosa cell tumor
Molecular findings
A total of five pathogenic or likely pathogenic mutations in FOXL2 (p.Cys134Trp), RB1 (p.Cys712Ter), TSC2 (c.2546-1G>C splice variant) and two mutations in TP53 gene (p.Arg273Cys and p.Leu257Arg ) were detected in the recurrent sex cord-stromal tumor. Only the FOXL2 mutation and one TP53 mutation (p.Leu257Arg) were also detected in the primary tumor with a low mutation allele frequency (MAF; 13% and 1%, respectively). Detailed list of the pathogenic or likely pathogenic variants is provided in Table 2.
Table 2. Detailed pathogenic or likely pathogenic variants detected in both tumors.
Primary AGCT
Recurrent AGCT
gene
genomic level
protein level
MAF (%)
MAF (%)
FOXL2
NM_023067.4:c.402C>G
p.Cys134Trp
13
46
RB1
NM_000321.2:c.2136C>A
p.Cys712Ter
not detected
87
TSC2
NM_000548.5:c.2546-1G>C
splice site
not detected
28
TP53
NM_000546.5:c.817C>T
p.Arg273Cys
1
45
TP53
NM_000546.5:c.770T>G
p.Leu257Arg
not detected
45
estimated tumor purity 80% estimated tumor purity 95%
Fig. 1. The representative morphological and immunohistochemical features of the tumors. A: Overview of the primary tumor (right) in close proximity to the structures of a mature cystic teratoma (left), 40x magnification (H&E staining). B: Histomorphology of the primary tumor, 100x magnification (H&E staining). C: Histomorphology of the recurrent tumor, 100x magnification (H&E staining). D: Histomorphology of the recurrent tumor (monstrous nuclei), 100x magnification (H&E staining). E: Aberrant p53 immunohistochemistry in the recurrent tumor, 100x magnification. F: Strong nuclear positivity of SF1 in the recurrent tumor, 100x magnification. 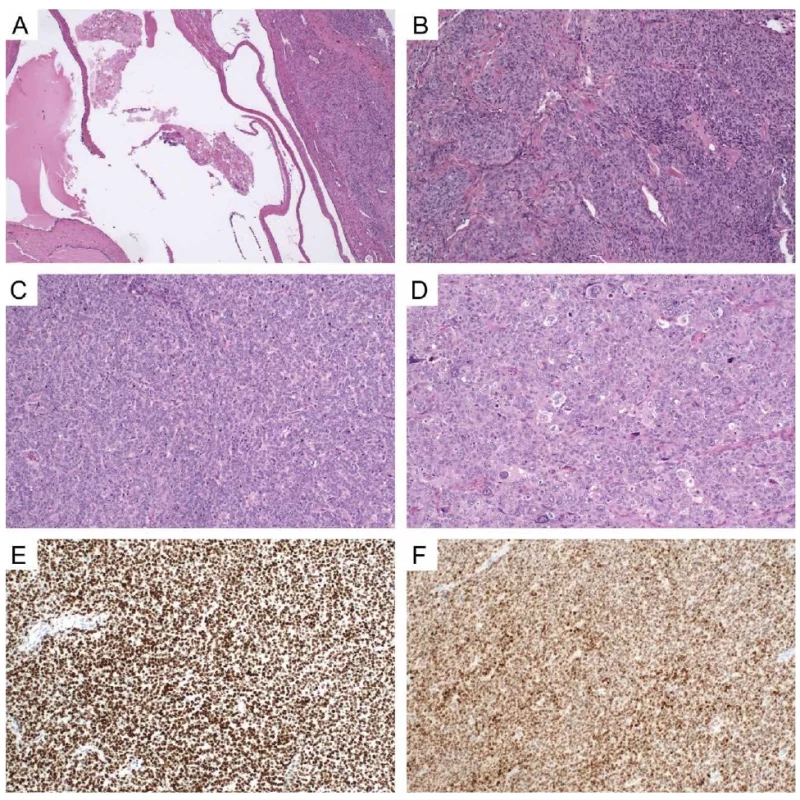
DISCUSSION
Sex cord-stromal tumors and mature teratoma developing within the same ovary simultaneously are exceedingly rare, with just over ten cases reported in the literature to date and the oldest dating back to 1919 (11). The difficulty lies in determining whether the tumor originates within the mature teratoma itself or develops independently as a synchronous neoplasm within the same ovary. This question can be comprehensively resolved through thorough sampling and the application of molecular pathology methods comparing the genetic landscape of both tumors. In most studies, the diagnosis was made solely based on morphological assessment using standard hematoxylin and eosin-stained slides, and only a few recent studies have incorporated immunohistochemistry into the diagnostic process (12-13). All previously documented cases have been diagnosed as granulosa cell tumors and no other type of sex cord-stromal tumor has been reported in this setting. We additionally provide a DNA NGS analysis to examine the molecular landscape of the primary and recurrent tumor. In our case, both the primary and recurrent tumor harbored the same missense FOXL2 mutation. This mutation serves as a diagnostic feature of AGCT as it is present in a majority of cases (4). However, this mutation can also be observed in a subset of SLCT and, rarely, in other sex cord-stromal tumors, highlighting the challenging differential diagnosis due to significant histomorphological overlap with AGCT (5-6,14-15). Additionally, a TP53 gene mutation was identified in the recurrent tumor, consistent with aberrant immunohistochemical p53 expression. Concurrent mutations of FOXL2 and TP53 have also been reported in other sex cord-stromal tumors, including sex cord-stromal tumor not otherwise specified (NOS) or gynandroblastoma (14-15). Regarding the molecular background of AGCT, Michálková et al. provided a complex analysis of 227 cases comparing primary and recurrent tumors and found RB1 mutation in one recurrent case in addition to the TP53, FOXL2, and TERT mutations. The TP53 mutation was detected in 3.5% of cases. However, in their cohort they did not detect any TSC2 mutations (16). The low MAF of the detected variants in the primary tumor suggests that these mutations are confined to a smaller subpopulation of cells within the primary tumor. In contrast, the additional mutations identified exclusively in the recurrent AGCT (TSC2, RB1, and second TP53 mutation) may represent secondary genetic events acquired during tumor progression, potentially explaining the increased aggressiveness observed in the recurrent tumor. In conclusion, we present the first molecularly characterized case of simultaneous ipsilateral ovarian AGCT and mature cystic teratoma with a late recurrence of the AGCT component. The tumor in both the primary and recurrent lesion harbored a missense FOXL2 and TP53 mutation typical for AGCT. TSC2 and RB1 mutations were present only in the recurrent tumor, indicating secondary mutations acquired during progression.
FUNDING
This work was supported by the Ministry of Health, Czech Republic (MH CZ DRO-VFN 64165 and AZV NU21-03-00238), by Charles University
(Project UNCE24/MED/018, SVV 260631), and by the European Regional Development Fund (EF16_013/0001674 and BBMRI_CZ LM2023033).
ACKNOWLEDGMENTS
The authors wish to extend their gratitude to Mgr. Zachary H. K. Kendall,
B.A. (Institute for History of Medicine and Foreign Languages, First Faculty of Medicine, Charles University in Prague) for the English language editing.
STATEMENTS AND DECLARATIONS
Competing Interests: The authors declare no competing financial and/or non-financial interests relevant to the content of this article.
Zdroje
- WHO Classification of Tumours Editorial Board. Female Genital Tumours. 5th ed. Lyon, France: International Agency for Research on Cancer (IARC); 2020.
- Schweppe KW, Beller FK. Clinical data of granulosa cell tumors. J Cancer Res Clin Oncol 1982; 104(1-2): 161-169.
- Babarović E, Franin I, Klarić M, et al. Adult Granulosa Cell Tumors of the Ovary: A Retrospective Study of 36 FIGO Stage I Cases with Emphasis on Prognostic Pathohistological Features. Anal Cell Pathol (Amst) 2018; 2018 : 9148124.
- Shah SP, Köbel M, Senz J, et al. Mutation of FOXL2 in granulosa-cell tumors of the ovary. N Engl J Med 2009; 360(26): 2719-2729.
- Karnezis AN, Wang Y, Keul J, et al. DICER1 and FOXL2 Mutation Status Correlates With Clinicopathologic Features in Ovarian Sertoli-Leydig Cell Tumors. Am J Surg Pathol 2019; 43(5): 628-638.
- Němejcová K, Hájková N, Krkavcová E, et al. A molecular and immunohistochemical study of 37 cases of ovarian Sertoli-Leydig cell tumor. Virchows Arch Published online November 27, 2024.
- Cong L, Wang S, Yeung SY, et al. Mature Cystic Teratoma: An Integrated Review. Int J Mol Sci 2023; 24(7): 6141.
- Gadducci A, Pistolesi S, Guerrieri ME, et al. Malignant Transformation in Mature Cystic Teratomas of the Ovary: Case Reports and Review of the Literature. Anticancer Res 2018; 38(6): 3669-3675.
- Moid FY, Jones RV. Granulosa cell tumor and mucinous cystadenoma arising in a mature cystic teratoma of the ovary: A unique case report and review of literature. Ann Diagn Pathol 2004; 8(2): 96-101.
- Dundr P, Bártů M, Bosse T, et al. Primary Mucinous Tumors of the Ovary: An Interobserver Reproducibility and Detailed Molecular Study Reveals Significant Overlap Between Diagnostic Categories. Mod Pathol 2023; 36(1): 100040.
- Silbermann E, Brody H. Granulosa cell tumor and dermoid cyst in same ovary. Am J Clin Pathol 1950.; 0(2): 177-183.
- Trivedi P, Patel T, Jain R, et al. Granulosa cell tumor arising in an ovary with mature teratoma. Indian J Pathol Microbiol 2009; 52(4): 559-560.
- Yoshino K, Fujita M, Shiki Y, et al. Granulosa cell tumor co-existing with a mature cystic teratoma of the ovary. J Obstet Gynaecol 2003; 23(3): 318-319.
- Stewart CJR, Amanuel B, De Kock L, et al. Evaluation of molecular analysis in challenging ovarian sex cord-stromal tumours: a review of 50 cases. Pathology 2020; 52(6): 686-693.
- Mayer R, Dandulakis M, Richards S, et al. Malignant Sex Cord-Stromal Tumor, Not Otherwise Specified, Harboring FOXL2, p53, and TERT Promoter Mutations: Report of a Case. Int J Gynecol Pathol 2020; 39(6): 567-572.
- Michálková R, Šafanda A, Hájková N, et al. The molecular landscape of 227 adult granulosa cell tumors of the ovary: Insights into the progression from primary to recurrence. Lab Invest Published online November 28, 2024.
Štítky
Patológia Súdne lekárstvo Toxikológia
Článok vyšiel v časopiseČesko-slovenská patologie

2025 Číslo 4-
Všetky články tohto čísla
- Patologie prsu – každodenní realita rutinní diagnostiky
- Svět molekulární biologie je paralelním vesmírem
- MONITOR aneb nemělo by vám uniknout, že...
- Papilární léze prsu: Diagnostická úskalí a přehled jednotek
- Imunohistochemické stanovení HER2 proteinu u karcinomů prsu – aktuální stav
- Vyšetřování prediktivních faktorů v léčbě karcinomu prsu
- Neobvyklý histopatologický nález v axilární uzlině pacientky s invazivním karcinomem NST: kazuistické sdělení a přehled literatury
- An unusual case of late recurrent adult granulosa cell tumor and mature teratoma arising within the same ovary, confirmed by NGS analysis
- Současné metody multiplexní imunohistochemie pro analýzu fixovaných tkáňových vzorků
- Česko-slovenská patologie
- Archív čísel
- Aktuálne číslo
- Informácie o časopise
Najčítanejšie v tomto čísle- Papilární léze prsu: Diagnostická úskalí a přehled jednotek
- Současné metody multiplexní imunohistochemie pro analýzu fixovaných tkáňových vzorků
- Patologie prsu – každodenní realita rutinní diagnostiky
- Imunohistochemické stanovení HER2 proteinu u karcinomů prsu – aktuální stav
Prihlásenie#ADS_BOTTOM_SCRIPTS#Zabudnuté hesloZadajte e-mailovú adresu, s ktorou ste vytvárali účet. Budú Vám na ňu zasielané informácie k nastaveniu nového hesla.
- Časopisy



