-
Články
- Časopisy
- Kurzy
- Témy
- Kongresy
- Videa
- Podcasty
- Kariéra
Ewing‘s sarcoma of the urinary bladder – the urologic and pathologic differential diagnosis and current therapeutic options
Ewingův sarkom močového měchýře – urologická a patologická diferenciální diagnostika a současné terapeutické možnosti
Východiska: Nádorová onemocnění močového měchýře se řadí na 11. místo v celosvětovém měřítku. Většina z nádorů se řadí k uroteliálním karcinomům, kdy méně běžné varianty (spinocelulární či adenokarcinomy) zahrnují zpravidla ≤ 10 % případů. Ostatní typy nádorů jsou výjimečné. Nález Ewingova sarkomu v močovém měchýři pak řadíme mezi raritní.
Případ: Prezentujeme případ 54leté pacientky vyšetřené pro bezbolestnou hematurii. V rámci došetření byl zjištěn objemný tumor močového měchýře, ale s ohledem na rozsah tumoru byla možná pouze diagnostická transuretrální resekce. Stadium onemocnění bylo dle primárního stagingu již vstupně pokročilé s metastatickým rozsevem, anemií a již zjištěnou obstrukcí horních močových cest.
Výsledky: Histologicky byl překvapivě v močovém měchýři prokázán Ewingův sarkom. Anemie způsobená hematurií a pokročilostí onemocnění byla korigována krevními převody a obstrukce pravé ledviny založením punkční nefrostomie. Navzdory velmi rychle stanovené diagnóze, dokončení stagingu a přípravě pacientky k další léčbě však pacientka umírá ještě před zahájením plánované systémové léčby.
Závěr: Diagnostika Ewingova sarkomu v měchýři je identická jako v případě ostatních typů měchýřových nádorů, tj. transuretrální resekce tumoru. V případě potvrzení tohoto histologického typu je nezbytné doplnit stagingová vyšetření a pokračovat multimodální léčbou. Zde hraje klíčovou roli včasně zahájená systémová chemoterapie, při vyloučení generalizace léčba zahrnuje také radikální cystektomii či radioterapii. Cílem našeho sdělení je prezentovat raritní případ tohoto onemocnění a poukázat na diferenciální diagnostiku, principy a možnosti léčby.
Klíčová slova:
terapie – Ewingův sarkom – karcinom měchýře
Authors: M. Král 1; D. Kurfúrstová 2; I. Hartmann 1; H. Študentová 3; J. Škarda 4
Authors place of work: Urologická klinika LF UP a FN Olomouc 1; Ústav klinické a molekulární patologie LF UP a FN Olomouc 2; Onkologická klinika LF UP a FN Olomouc 3; Ústav klinické a molekulární patologie a lékařské genetiky, LF OU a FN Ostrava 4
Published in the journal: Klin Onkol 2023; 36(4): 314-319
Category: Kazuistiky
Summary
Background: Bladder cancer is 11th most common cancer worldwide. Histologically, most of the tumors are classified as urothelial carcinomas. Less common variants (squamous cell or adenocarcinomas) usually comprise up to 10% of cases. Other types of tumors are exceptional. The finding of Ewing‘s sarcoma in the bladder is considered extremely rare.
Case: We present the case of a 54-year-old female patient examined for painless hematuria. During the follow-up examination, a bulky tumor of the bladder was detected, but considering the extent of the bladder tumor, only a diagnostic transurethral resection was possible. According to the primary staging, the disease was already advanced at the time of admission with metastatic spread, anemia and present obstruction of the upper urinary tract.
Results: Histologically, Ewing‘s sarcoma was surprisingly demonstrated in the urinary bladder. Anemia caused by hematuria and advanced disease was corrected by blood transfusions and obstruction of the right kidney by puncture nephrostomy. However, despite a very quick diagnosis, completion of staging and preparation of the patient for further treatment, the patient had died before the planned systemic treatment began.
Conclusion: The diagnosis of Ewing‘s sarcoma is identical to that of the other bladder tumors, i.e. transurethral resection. In the case of confirmation of this histological type, it is necessary to complete staging examinations and start multimodal treatment. Early systemic chemotherapy plays a key role and if metastatic spread is excluded, radical cystectomy or radiotherapy are included, too. The aim of our communication is to present a rare case of this disease, discuss the differential diagnosis and point out the principles and possibilities of its treatment.
Keywords:
Bladder cancer – treatment – Ewing‘s Sarcoma
Introduction
Bladder cancer (BC) is the most common malignancy affecting the urinary system. It is the 11th most common cancer worldwide. In Europe and USA, urothelial cancer is the predominant histologic type of bladder cancer and it accounts approx. for 90% of all cases. Squamous cell carcinoma, adenocarcinoma rising from urachus and “non-urachal” adenocarcinoma, small cell carcinoma and metastatic tumors of the bladder are the rest of histologic types of tumors. Other types of cancers can be seen seldom only. Therefore, finding Ewing‘s sarcoma in the urinary bladder is extremely rare. So far, only 21 cases have been presented in the literature. Standard treatment of the urothelial type of BC is transurethral resection of the tumor, usually followed by intravesical chemotherapy instillation. If bladder muscle invasion is found, radical cystectomy is indicated (with or without neoadjuvant intravenous chemotherapy). Finding a different histological type of bladder cancer requires a specific approach depending on the histology. Especially in Ewing‘s sarcoma, the treatment must be as urgent and radical as possible. The aim of this case report is to present a patient with Ewing‘s sarcoma of the urinary bladder, its diagnostics and therapy with emphasis on the extremely unfavorable course of this disease when the diagnosis is made late.
Case report
We present a case report of a 54-year-old woman examined by a local urologist due to gross hematuria and right side flank pain. From the first presentation, the patient had general symptoms, mainly fatigue, nausea, fever and loss of weight. In the laboratory tests, anemia (hemoglobin 93 g/L), leukocytosis (16,1×109/L), mild renal insufficiency (serum creatinine 173 µmol/L) with decrease of renal clearance (0,43 mL/s/1,73 m2) were found. The abdominal sonography revealed secondary obstruction of the right kidney due to mass in the urinary bladder. The out-patient cystoscopy was performed and a huge, almost completely urinary bladder filling, necrotic tumor of 7 cm in diameter was identified with right ureteral ostium blockade. Preoperative chest X-ray showed suspicion for small pulmonary metastasis on the right side. CT of the abdomen proved huge intravesical formation with enlarged pelvic lymph nodes. Bimanual palpation under general anesthesia revealed bulky mass in the suprapubic area. Subsequent transurethral bladder tumor resection (TURBT) was performed but only with a diagnostic aim due to the massive extension of the tumor. Because of renal insufficiency and right side flank pain due to kidney obstruction, the right side nephrostomy in supine position was placed. The patient was discharged 2 days later.
Multiple tissue samples of 3–7 mm in size were sent to pathology and after basic processing and embedding in paraffin blocks, histological slides stained with basic hematoxylin-eosin staining were made. Microscopically, it was a focally necrotic multicellular tumor consisting of cells with oval nuclei and a minimal amount of cytoplasm. In the immunohistochemical examination, these cells diffusely expressed vimentin, CD99, NSE and focally EMA (Fig. 1). Negative immunohistochemical markers included S-100, CK18, chromogranin, CK20, CK7, LCA, synaptophysin, CKV, CD56, CD20, CD3, AE1 and AE3. The samples were further subjected to a cytogenetic examination using the fluo - rescence in situ hybridization (FISH) method using the dual color break apart probe, which demonstrated an aberration of EWSR1 gene in 34% of the nuclei. The diagnosis of Ewing‘s sarcoma was therefore established.
Postoperatively, fluorodeoxyglucose PET/CT scan was performed with multiple pathologic lesions detected: in locally advanced bladder tumor infiltrating the vagina with multiple pelvic and mediastinal lymphadenopathy, in the right liver lobe, right adrenal gland and multiple lung nodules (Fig. 2). Multidisciplinary team (urologist, medical oncologist, radiation oncologist, pathologist and radiologist) recommended prompt administration of chemotherapy. Unfortunately, 2 weeks after the primary transurethral resection, general status worsening with metabolic alteration occurred (hyponatremia 123 mmol/L, hyperkalemia 7,03 mmol/L, hypochloremia 88 mmol/L, creatinine 219 µmol/L, metabolic acidosis and liver test dysfunction with ALT 2,46 µkat/L and AST 10,53 µkat/L). Despite intensive care and metabolic correction, the overall clinical status gradually worsened and the patient died within 2 days, without chemotherapy being initiated. The autopsy revealed advanced Ewing‘s sarcoma having been spread into the perivesical soft tissues and the vaginal wall. As expected according to PET/CT, multiple metastases of the sarcoma to the lungs, liver and adrenal glands were detected. Thromboembolism to the branches of the pulmonary artery was identified as the potential immediate cause of death.
Fig. 1. Fragments of a tumor consisting of medium-sized cells with large hyperchromatic nuclei, coarse chromatin structure with abundant mitotic activity (A); immunohistochemistry, tumor cells express vimentin (B); CD99 (C); neuron specifi c enolase (scale bar 100 μm) (D). 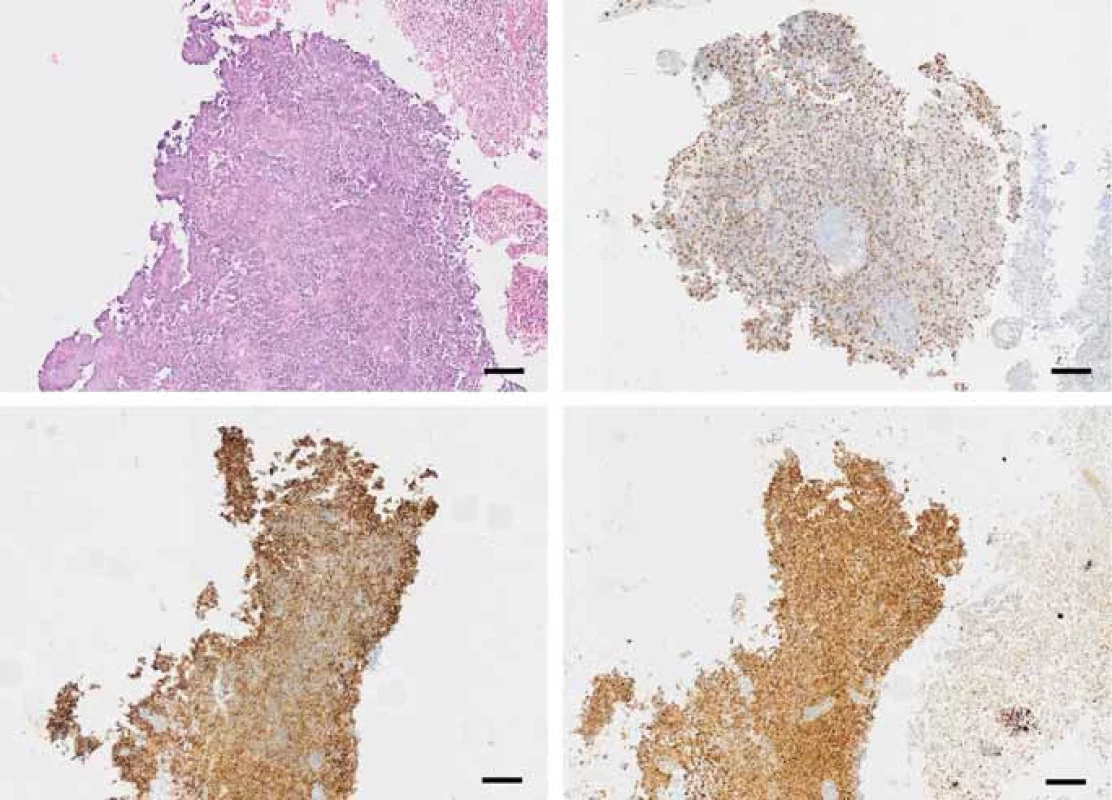
Fig. 2. FDG-PET/CT scan (due to renal deterioration without iodine contrast i.v. Axial plane, non-contrast CT showing huge urinary bladder mass (A); axial plane, FDG accumulating in tumorous bladder tissue (non-accumulating necrotic inner part of the tumor) (B); coronal plane, obstructed right kidney due to bladder mass (C); FDG accumulation lesion in the lower lobe of the left lung (D). 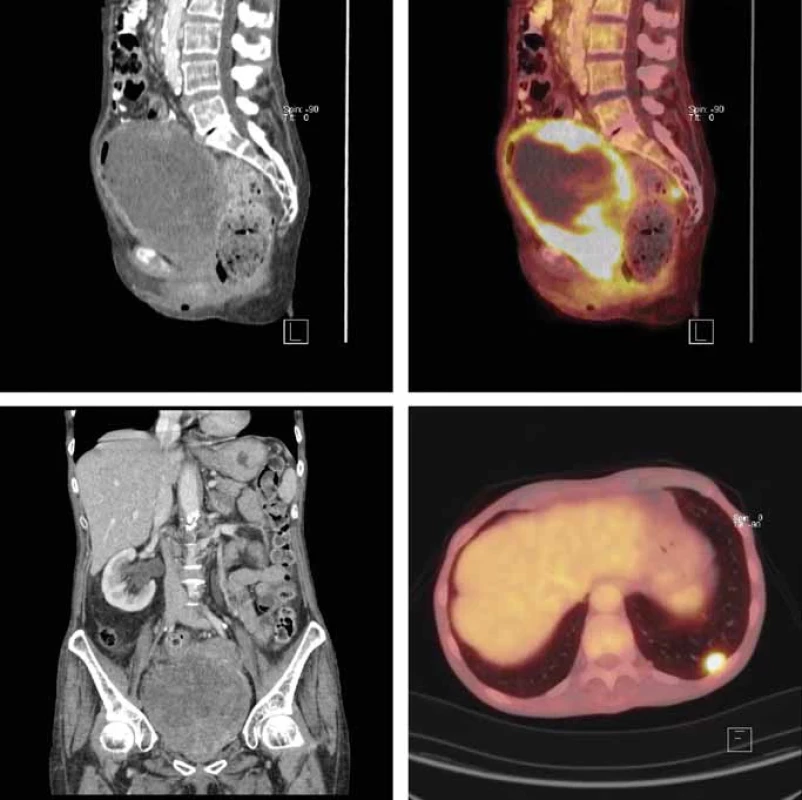
FDG – fl uorodeoxyglucose Discussion
Following multiple myeloma and osteosarcoma, ES is the third most common primary bone malignancy. The highest prevalence is in Western Europe, USA and New Zealand with an incidence of three cases per million per year. Males are affected more often. Ewing‘s sarcoma is very rare in African and Asian populations. Although this type of tumor was first described by James Ewing as early as in 1921, other (initially separate) nosological units were gradually assigned to it: primitive peripheral neuroectodermal tumors (PNET), Askin‘s tumor of the chest wall, and extraskeletal Ewing‘s sarcoma (EES), diagnosed in 1969. A group called the Ewing‘s sarcoma family (ESF) was thus created. Currently, the name Ewing‘s sarcoma is preferred according to WHO Classification of Tumors Editorial Board (Lyon, France 2020). It is typical for Ewing‘s sarcoma that it arises most often in flat or long bones, but it can arise in any skeletal or extraskeletal soft tissue. ES is the second most common bone malignancy in children and adolescents. In up to 80% of cases, it affects people aged < 20 years and rarely occurs in people over the age of 30 [1–3]. Ewing‘s sarcomas are characterized by a high malignant potential, and early hematogenous dissemination, when dissemination may occur in extraosseous tissues, e. g. in the liver, lungs, pancreas, testicles, uterus, kidney, bladder or rectum. However, these organs can rarely be the origin of isolated primary extraskeletal Ewing‘s sarcomas [4–7]. Compared to common skeletal Ewing‘s sarcoma lesions, extraskeletal Ewing‘s sarcomas are present in older patients (mean age of 27.5 years in extra-osseous tumor vs. 16.8 years in osseous tumor) [8]. This type of the tumor is more frequent in women.
To the date of our article submission, 21 presented cases of primitive ES in the urinary bladder were found in the available literature [9]. Compared to common urothelial bladder cancer patients, bladder Ewing‘s sarcoma patients tend to be significantly younger (> 50% of patients were at the age of < 50 years, often < 30 years old). The symptomatology of these patients was usually identical to common urothelial tumors: the most common was painless gross hematuria, a bulky intravesical mass or extensive lymphadenopathy, secondary megaureters and lymphedema of the lower extremities were also present. The systemic clinical symptoms were similar to other bladder tumors such as fatigue, loss of weight, sweating or fever of unknown origin and were present in 20% of ES patients [5,10]. From a histologic point of view, these tumors consist of small round cells, which are characterized by gene rearrangement due to the formation of FET-ETS fusion genes. The most common fusion gene found in ES cells is EWSR1-FLI1, the second most common is EWSR1-ERG. Other mutations are rare. The detection of one of these fusion genes is essential for the diagnosis of ES. Other mutations that can be demonstrated include CDKN2A, TP53 or STAG2 genes [11–14].
As a diagnostic tool, cystoscopy and transurethral resection of the tumor play a key role. CT urography is the essential imaging method [15]. In the majority of cases, evidence of Ewing‘s sarcoma is a surprising finding in histology, and therefore it is necessary to perform FDG-PET/CT (head to toe) to assess the extent of the disease to plan the type and sequence of treatment steps. Compared to common urothelial bladder tumors, ES tumors are primarily characterized by large tumor masses (often 6–12, but even more centimeters in diameter). Therefore, complete TURBT is usually impossible.
If ES is diagnosed (by bladder biopsy or TURBT) and metastatic disease is ruled out, it is recommended to start with multiagent chemotherapy for at least 9 weeks prior to surgery with the aim to downstage the tumor and enhance the probability of achieving a complete resection (CR) with microscopically negative margins. After restaging imaging methods are performed and no metastasis are found, a radical treatment is the next step. Following surgical resection, adjuvant chemotherapy should be offered because it improves relaps-free survival and overall survival (OS) in a majority of patients. This path is supported by the fact of high aggressiveness of ES and the frequent presence of clinically undetectable metastases already in the early phase of the disease. The studies below also support this theory. Similarly to other Ewing‘s sarcomas sites, for better local control, the postoperative radiotherapy is often a recommended option [16]. At this point, the role of the pathologist should be emphasized, not only in terms of accurate histological classification, but also the speed of its determination to ensure timely systemic treatment: if radical chemotherapy is not administered within a few weeks after the diagnosis is made, the metastases do occur and the patient dies. Systemic chemotherapy is crucial for the management of all Ewing‘s sarcomas. Although the metastatic disease is only present in 25% of newly diagnosed patients, up to 90% of patients relapse if systemic chemotherapy is not applied. Univariate and multivariate analysis showed that unfavorable factors for event-free survival (EFS) and OS include tumor size > 8 cm, high LDH value (high tumor burden indicator), positive surgical margins, poor response to chemotherapy and primarily metastatic stage [17].
Due to small numbers of bladder Ewing‘s sarcomas, so far it has not been possible to establish definitive guidelines regarding its management and treatment. Therefore, our experience is based and extrapolated from skeletal and extraskeletal Ewing‘s sarcomas. Analysis of data from patients with extraskeletal Ewing sarcomas showed that truncal location, upfront surgery and positive surgical margins led to lower event free survival and overall survival. Therefore, if it is not possible to ensure radical extirpation of the tumor with negative surgical margins, primary chemotherapy is clearly recommended, and only in case downstaging is achieved, it is recommended to continue with surgical treatment. In the event that surgical treatment is not radical (or leads to significant morbidity), radical (definitive) radiotherapy can be considered [18–21]. The approach to the systemic treatment of ES depends to a great extent on whether the disease is localized or metastatic.
Localized disease
In this case, it is a combination of systemic and local treatment. In terms of systemic treatment, multiagent chemotherapy VDC/IE (vincristine, doxorubicin, cyclophosphamide and ifosfamide/etoposide) or VIDE (vincristine, ifosfamide, doxorubicin, etoposide) regimens are used nowadays. Randomized trials showed that the combination of VDC/IE resulted in improved OS with shorter treatment duration and lower toxicity. The combination treatment of VDC/IE is based on the data of the Intergroup Ewing sarcoma studies (IESS I–III) and is the preferred treatment in the US, while the combination of VIDE is commonly applied in Europe according to the data of the studies Euro-E.W.I.N.G.99 and Ewing-2008. Due to hematological toxicity, hematological growth factors (e. g. filgrastim) are often included in the combination scheme. The advantages of administering the VDC/IE combination compared to VIDE were confirmed in a phase III study (EE2012) [22,23]. The duration of individual cycles is usually 3 weeks, but for example in younger patients (< 18 years) the so-called interval-compressed VDC/IE regimens lasts 2 weeks. The duration of intervals (2 vs. 3 weeks) was tested in a clinical trial which confirmed superiority of interval-compressed chemotherapy in localized ES showing improved EFS and OS. This was confirmed for both groups of patients regardless of age (< 18 years and ≥ 18 years), although patients aged > 18 years with primarily pelvic localization of the tumor and poor histologic response (< 90% necrosis in the tumor after neoadjuvant treatment) were among those at high risk for disease relapse [24]. Following neoadjuvant chemotherapy and restaging examinations, radical treatment is indicated – surgery, radiotherapy or combination, taking into account the primary location and disease extent.
Metastatic disease
Patients with primarily metastatic dis - ease form a group with a very adverse prognosis. The purpose of treatment is to prolong OS, delay progression and alleviate symptoms of local tumor progression, presented usually by pain. Metastatic Ewing sarcomas comprise a very heterogeneous group of different primary locations and extent of disease. Moreover, they account for only 25–30% of all ES cases, so there are not enough randomized studies to allow a more extensive analysis of these cases. Therefore, there is general agreement that all new patients should be offered participation in clinical trials to assess new treatment strategies. If a clinical trial participation is not feasible, these patients are treated with identical chemotherapy regimens as for localized ES [25,26].
Conclusion
Ewing‘s sarcoma of the urinary bladder is an extremely rare unit with similar symptomatology to the typical urothelial bladder cancer. Unfortunately, its clinical course is usually unfavourable and the prognosis is extremely poor. The therapy consists of early and maximally radical surgical treatment (radical cystectomy and urine derivation), optimally with both radiation and chemotherapy even in non-metastatic cases. The prognosis of Ewing‘s sarcoma of the urinary bladder is similar to both skeletal and other extraskeletal locations depending on primary disease extent, age of the patient, technical radicality of surgical treatment and multimodal approach.
Zdroje
1. Choi JH, Ro JY. The 2020 WHO classification of tumors of soft tissue: selected changes and new entities. Adv Anat Pathol 2021; 28 (1): 44–58. doi: 10.1097/PAP.000000000 0000284.
2. Cotterill SJ, Ahrens S, Paulussen M et al. Prognostic factors in Ewing‘s tumor of bone: analysis of 975 patients from European Intergroup Cooperative Ewing‘s Sarcoma Study Group. J Clin Oncol 2000; 18 (17): 3108–3114. doi: 10.1200/JCO.2000.18.17.3108.
3. Bernstein M, Kovar H, Paulussen M et al. Ewing‘s sarcoma family of tumors: current management. Oncologist 2006; 11 (5): 503–519. doi: 10.1634/theoncologist.11-5-503.
4. Bleyer WA, O’Leary M, Barr R et al. Cancer epidemiology in older adolescents and young adults 15 to 29 years of age, including SEER incidence and survival: 1975–2000. [online]. Available from: https: //seer.cancer.gov/archive/publications/aya/aya_mono_complete.pdf.
5. Tonyalı Ş, Yazıcı S, Yeşilırmak A et al. The Ewing‘s sarcoma family of tumors of urinary bladder: a case report and review of the literature. Balkan Med J 2016; 33 (4): 462–466. doi: 10.5152/balkanmedj.2016.16533.
6. Zhang Y, Nong W, Ren Y et al. Ewing‘s sarcoma of the cervix: a case report and review of literature. Histol Histopathol 2020; 35 (5): 475–480. doi: 10.14670/HH-18-181.
7. Hu X, Li D, Cai J. Experience of CT diagnosis and management of primary renal Ewing‘s sarcoma: a retrospective analysis of 6 cases and a literature review. Medicine (Baltimore) 2022; 101 (49): e32189. doi: 10.1097/MD.0000 000000032189.
8. Pradhan A, Grimer RJ, Spooner D et al. Oncological outcomes of patients with Ewing‘s sarcoma: is there a difference between skeletal and extra-skeletal Ewing‘s sarcoma? J Bone Joint Surg Br 2011; 93 (4): 531–536. doi: 10.1302/0301-620X.93B4.25510.
9. Kassab NB, Ballester PSH, Sanz PS et al. Ewing-like sarcoma bladder primary tumour: a case report and literature review. Urol Case Rep 2022; 44 : 102139. doi: 10.1016/j.eucr.2022.102139.
10. Rud NP, Reiman HM, Pritchard DJ et al. Extraosseous Ewing‘s sarcoma. A study of 42 cases. Cancer 1989; 64 (7): 1548–1553. doi: 10.1002/1097-0142 (19891001) 64 : 7<1548:: aid-cncr2820640733>3.0.co; 2-w.
11. Tirode F, Surdez D, Ma X et al. Genomic landscape of Ewing sarcoma defines an aggressive subtype with co-association of STAG2 and TP53 mutations. Cancer Discov 2014; 4 (11): 1342–1353. doi: 10.1158/2159-8290.CD-14-0622.
12. Lerman DM, Monument MJ, McIlvaine E et al. Tumoral TP53 and/or CDKN2A alterations are not reliable prognostic biomarkers in patients with localized Ewing sarcoma: a report from the Children‘s Oncology Group. Pediatr Blood Cancer 2015; 62 (5): 759–765. doi: 10.1002/pbc.25340.
13. Jo VY. EWSR1 fusions: Ewing sarcoma and beyond. Cancer Cytopathol 2020; 128 (4): 229–231. doi: 10.1002/cncy.22239.
14. Gorthi A, Bishop AJR. Ewing sarcoma fusion oncogene: at the crossroads of transcription and DNA damage response. Mol Cell Oncol 2018; 5 (4): e1465014. doi: 10.1080/23723556.2018.1465014.
15. Babjuk M, Böhle A, Burger M et al. EAU guidelines on non-muscle-invasive bladder cancer. [online]. Available from: https: //uroweb.org/guideline/non-muscle-invasive-bladder-cancer/.
16. Schuck A, Ahrens S, Paulussen M et al. Local therapy in localized Ewing tumors: results of 1058 patients treated in the CESS 81, CESS 86, and EICESS 92 trials. Int J Radiat Oncol Biol Phys 2003; 55 (1): 168–177. doi: 10.1016/s0360-3016 (02) 03797-5.
17. Tural D, Molinas Mandel N, Dervisoglu S et al. Extraskeletal Ewing‘s sarcoma family of tumors in adults: prognostic factors and clinical outcome. Jpn J Clin Oncol 2012; 42 (5): 420–426. doi: 10.1093/jjco/hys027.
18. Mathew J, Arjunan R, Dasappa A et al. Prognostic factors and clinical outcomes in extraskeletal Ewing sarcoma: a cohort study. Ann Surg Oncol 2023; 30 (5): 3084–3094. doi: 10.1245/s10434-022-12992-1.
19. Cash T, McIlvaine E, Krailo MD et al. Comparison of clinical features and outcomes in patients with extraskeletal versus skeletal localized Ewing sarcoma: a report from the Children‘s Oncology Group. Pediatr Blood Cancer 2016; 63 (10): 1771–1779. doi: 10.1002/pbc.26096.
20. Lynch AD, Gani F, Meyer CF et al. Extraskeletal versus skeletal Ewing sarcoma in the adult population: controversies in care. Surg Oncol 2018; 27 (3): 373–379. doi: 10.1016/j.suronc.2018.05.016.
21. Abboud A, Masrouha K, Saliba M et al. Extraskeletal Ewing sarcoma: diagnosis, management and prognosis. Oncol Lett 2021; 21 (5): 354. doi: 10.3892/ol.2021.12 615.
22. Brennan B, Kirton L, Marec-Bérard P et al. Comparison of two chemotherapy regimens in patients with newly diagnosed Ewing sarcoma (EE2012): an open-label, randomised, phase 3 trial. Lancet 2022; 400 (10362): 1513–1521. doi: 10.1016/S0140-6736 (22) 01790-1.
23. Whelan J, Le Deley MC, Dirksen U et al. High-dose chemotherapy and blood autologous stem-cell rescue compared with standard chemotherapy in localized high-risk Ewing sarcoma: results of Euro-E.W.I.N.G.99 and Ewing-2008. J Clin Oncol 2018; 36 (31): JCO2018782516. doi: 10.1200/JCO.2018.78.2516.
24. Cash T, Krailo MD, Buxton A et al. Long-term outcomes in patients with localized Ewing sarcoma treated with interval-compressed chemotherapy: a long-term follow-up report from Children’s Oncology Group study AEWS0031. J Clin Oncol 2022; 40 (16 Suppl): 11505–11505.
25. Haveman LM, van Ewijk R, van Dalen EC et al. High-dose chemotherapy followed by autologous haematopoietic cell transplantation for children, adolescents, and young adults with primary metastatic Ewing sarcoma. Cochrane Database Syst Rev 2021; 9 (9): CD011405. doi: 10.1002/14651858.CD011405.pub2.
26. Pinkerton CR, Bataillard A, Guillo S et al. Treatment strategies for metastatic Ewing‘s sarcoma. Eur J Cancer 2001; 37 (11): 1338–1344. doi: 10.1016/s0959-8049 (01) 00131-9.Štítky
Detská onkológia Chirurgia všeobecná Onkológia
Článok vyšiel v časopiseKlinická onkologie
Najčítanejšie tento týždeň
2023 Číslo 4- Spasmolytický účinek metamizolu
- Brno opět přivítá onkology a nelékařské zdravotnické pracovníky
- I „pouhé“ doporučení znamená velkou pomoc. Nasměrujte své pacienty pod křídla Dobrých andělů
- Realita liečby bolesti v paliatívnej starostlivosti v Nemecku
- MUDr. Lenka Klimešová: Multiodborová vizita je kľúč k efektívnejšej perioperačnej liečbe chronickej bolesti
-
Všetky články tohto čísla
- Pět P tuzemské onkologie – pochvala, podpora, polemika, pochybnosti, poklesky
- Novinky v piatej edícii klasifikácie nádorov semenníkov podľa Svetovej zdravotníckej organizácie
- Volná cirkulující DNA a její potenciál v diagnostice a léčbě maligních lymfomů
- Study of KDM1A and VEGF changes as the responsible genes in the angiogenesis of breast cancer
- Neurobiológia mnohopočetného myelómu a jej terapeutické využitie – výsledky pilotnej štúdie s kontrolným ramenom
- Enzalutamid a abirateron v léčbě pacientů s metastatickým kastračně rezistentním karcinomem prostaty po podání chemoterapie
- Jak nám mohou pacienti pomoci být ještě lepšími lékaři – edukační leták „Než půjdu k lékaři“
- Ewing‘s sarcoma of the urinary bladder – the urologic and pathologic differential diagnosis and current therapeutic options
- Léčba Castlemanovy choroby siltuximabem – popis případu a přehled literatury
- Aktuality z odborného tisku
- Nová učebnice ukazuje, jak může obecný paliativní přístup významně zlepšit nemocniční péči o pacienty se závažnými diagnózami i jejich blízké
- Klinická onkologie
- Archív čísel
- Aktuálne číslo
- Informácie o časopise
Najčítanejšie v tomto čísle- Neurobiológia mnohopočetného myelómu a jej terapeutické využitie – výsledky pilotnej štúdie s kontrolným ramenom
- Jak nám mohou pacienti pomoci být ještě lepšími lékaři – edukační leták „Než půjdu k lékaři“
- Enzalutamid a abirateron v léčbě pacientů s metastatickým kastračně rezistentním karcinomem prostaty po podání chemoterapie
- Ewing‘s sarcoma of the urinary bladder – the urologic and pathologic differential diagnosis and current therapeutic options
Prihlásenie#ADS_BOTTOM_SCRIPTS#Zabudnuté hesloZadajte e-mailovú adresu, s ktorou ste vytvárali účet. Budú Vám na ňu zasielané informácie k nastaveniu nového hesla.
- Časopisy



