Second recurrence of breast cancer 36 years after initial surgery and 18 years after the first recurrence – an extremely rare case
Authors:
Y. Maezawa 1; T. Terada 2; H. Kobayashi 2; G. Ohara 1; T. Saito 3; N. Takayashiki 4; H. Satoh 1
Authors‘ workplace:
Department of Respiratory Medicine, Mito Kyodo General Hospital, Mito, Japan
1; Department of Internal Medicine, Mito Kyodo General Hospital, Mito, Japan
2; Department of Breast Surgery, Mito Kyodo General Hospital, Mito, Japan
3; Department of Pathology, Mito Kyodo General Hospital, Mito, Japan
4
Published in:
Klin Onkol 2026; 39(2): 117-121
Category:
Case Report
doi:
https://doi.org/10.48095/ccko2026117
Overview
Background: Breast cancer is a one of the most common causes of cancer death in the world. Late recurrence is a notable characteristic of this disease. Case: We experienced an extremely rare case of breast cancer that developed a second recurrence 36 years after the initial surgery and 18 years after the first recurrence, manifesting as bilateral pleural effusion and multiple bone metastases. Conclusion: This report highlights that it is important for both patients and doctors to be aware that such patients exist.
Keywords:
breast cancer – late recurrence – second recurrence
Introduction
Breast cancer ranks second in global cancer prevalence after lung cancer and fourth in cancer-related mortality after lung, colorectal, and liver cancers [1]. With particular attention to cancers that occur in women, it is the most frequently diagnosed malignancy, making its epidemiology particularly important.
In surveillance for breast cancer, recommendations for how many years to perform mammograms are unclear. Some reports have suggested that annual mammography is a useful approach [2], although the optimal duration of such surveillance has not been established. The history of breast cancer is itself a risk factor for the development of secondary breast cancer [3], which has been reported to increase the incidence of distant metastases and mortality [4,5]. Consequently, additional imaging modalities such as ultrasonography or MRI are sometimes employed, although considerable variation exists among patients’ staging, treatment, and pathological findings.
Herein, we present a rare case of right breast cancer that recurred 18 years after first recurrence and 36 years after initial surgery, manifesting as bilateral pleural effusion and multiple bone metastases.
Case report
An 80-year-old woman, who had been followed by her family physician for hypertension and dyslipidemia, was referred to our hospital due to bilateral pleural effusion.
Thirty-six years ago, the patient underwent a right total mastectomy with axillary lymph node dissection for right breast cancer at a hospital other than ours. She was followed for 15 years without recurrence and was subsequently discharged from follow-up. Eighteen years after surgery, she visited the hospital because of right upper limb edema and was diagnosed with right axillary lymph node recurrence. At that time, information regarding pathological findings, including immunohistochemistry, was not available at the hospital. The tumor was presumed to be hormone receptor-positive based on the extremely late recurrence. She was treated with tamoxifen and concurrent radiotherapy (50 Gy in 25 fractions). After eight years of tamoxifen therapy, the medication was switched to anastrozole, which was continued for an additional five years. Since there was no evidence of recurrence, both endocrine therapy and follow-up were discontinued.
The patient presented to family physician with enlarged right supraclavicular lymph nodes 5 months previously and cervical lymph nodes 1 month before previously. Around the same time, a chest X-ray performed by her local physician revealed bilateral pleural effusion. Diuretic therapy was initiated for presumed heart failure, based on an elevated brain natriuretic peptide (BNP) level of 157 pg/mL, but it was not effective. She was then referred to our hospital for further evaluation and management of the pleural effusion.
Upon presentation to our outpatient clinic, the patient was alert. Her vital signs were as follows: blood pressure, 137/83 mmHg; pulse rate, 81 beats/min; respiratory rate, 24 breaths/min; oxygen saturation, 94% on room air; and body temperature 36.9 °C.
Physical examination revealed decreased breath sounds in both lung fields, pitting edema of both lower extremities, and enlarged lymph nodes in the right clavicular and cervical and left axillary regions.
Laboratory tests showed no elevation of inflammatory markers, and the BNP level was 112.8 pg/mL, consistent with the findings reported by her previous physician. Blood tests also revealed elevated tumor markers, with a carcinoembryonic antigen (CEA) level of 11.8 ng/mL and a cancer antigen 15-3 (CA15-3) level of 56.4 U/mL.
Chest radiography demonstrated bilateral pleural effusion (Fig. 1). Contrast-enhanced CT of the chest and abdomen revealed enlarged lymph nodes in the cervical and right supraclavicular regions, as well as para-aortic lymphadenopathy in abdomen. Passive atelectasis was observed, but there were no apparent lung masses, and no enlargement of hilar or mediastinal lymph nodes was noted.
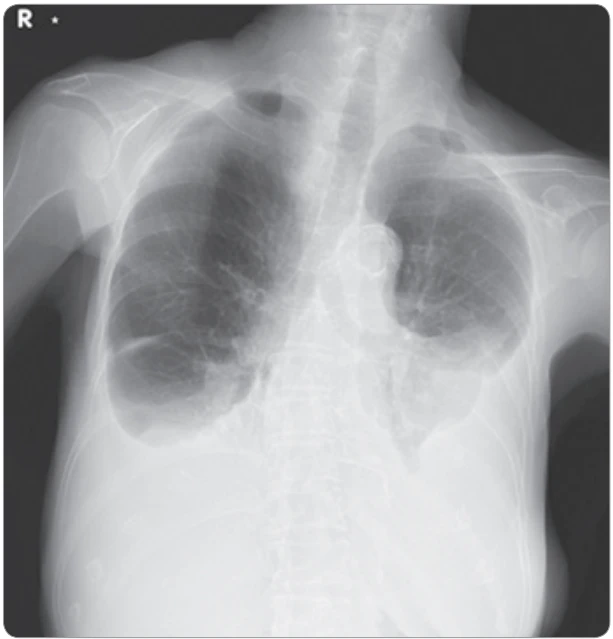
Transthoracic echocardiography revealed no evidence of heart failure. Right-sided thoracentesis yielded yellowish, clear, exudative pleural fluid. Cytological examination revealed class IV findings, suggestive of adenocarcinoma. Two weeks later, at a follow-up outpatient visit, progression of the pleural effusion was noted, prompting repeat thoracentesis and preparation of the pleural effusion-cell block.
Whole-body evaluation with PET-CT revealed lymphadenopathy with fluorodeoxyglucose (FDG) uptake in the right cervical, right supraclavicular, and left axillary regions (maximum standardized uptake value (SUVmax) 6.67 in the right supraclavicular region). No mass lesion suspicious for lung cancer was identified (Fig. 2). Bilateral pleural effusion and ascites were also present. Multiple areas of FDG uptake were identified in the thoracic spine, right acetabulum, and right femoral greater trochanter, raising suspicion for bone metastases. No abnormal findings were detected in the liver, biliary system, pancreas, kidneys, or spleen.
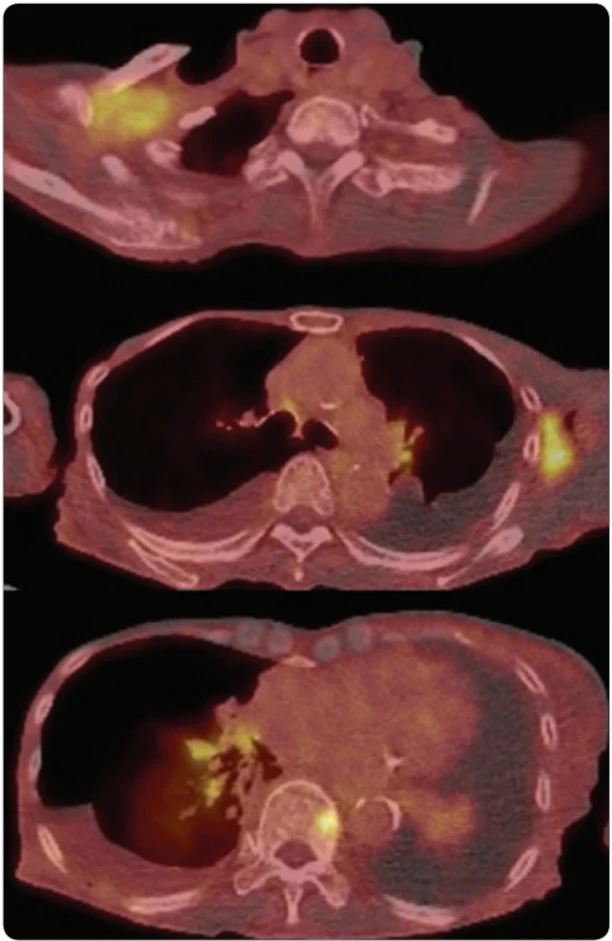
Although an outpatient appointment had been scheduled to explain the results, the patient was transported to the emergency department before the scheduled visit due to worsening dyspnea. She was diagnosed with carcinomatous pleuritis, and a chest tube was inserted into the right pleural cavity, followed by hospital admission.
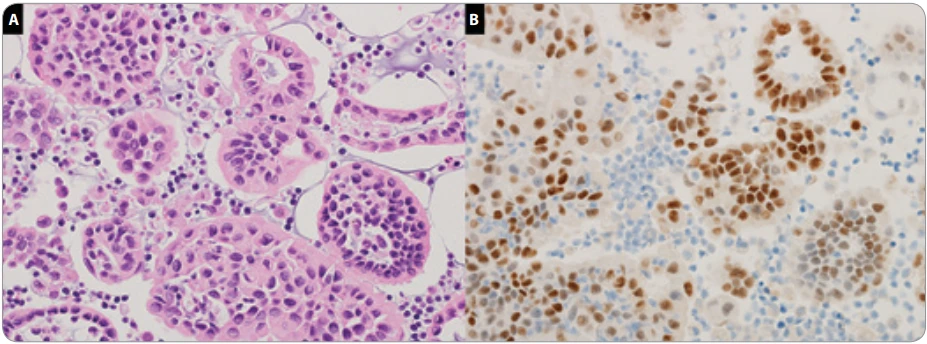
Pathological examination of the pleural effusion cell block revealed columnar atypical cells with enlarged and irregular nuclei and pale eosinophilic cytoplasm proliferating in an acinar pattern. Periodic acid–Schiff (PAS) stain showed faint intracytoplasmic positivity. Immunohistochemical staining showed positivity for cytokeratin-7 (CK7), estrogen receptor (ER), progesterone receptor (PgR), and gross cystic disease fluid protein-15 (GCDFP-15), negativity for cytokeratin-20 (CK20) and thyroid transcription factor 1 (TTF-1) (Fig. 3). The pathological findings were consistent with metastatic breast carcinoma. PET-CT showed no abnormal FDG uptake in the left breast, supporting the diagnosis of postoperative recurrence of right breast cancer. Although initiation of chemotherapy was considered, her poor general condition, worsening oxygenation, and progressive dyspnea led to a best supportive care approach. The patient died on the eighth day of hospitalization.
Discussion
Postoperative recurrence of breast cancer most frequently occurs within 5 years after surgery, particularly within the first 1–2 years [6]. Reports of recurrence developed more than 30 years after the initial diagnosis are extremely uncommon [7–11]. Metastatic breast cancer may present with either solitary or diffuse metastatic lesions and can follow various clinical courses [12]. However, most reported cases of recurrence after an interval exceeding 30 years have involved solitary or limited numbers of metastatic sites [7–11], and reports of cases, such as ours, with diffuse metastases throughout the body remain limited. It has been reported that second recurrences of breast cancer usually occur within five years after the first recurrence [13,14]. Therefore, a case such as ours, in which the second recurrence developed 18 years after the first recurrence and 36 years after the initial treatment, is considered extremely rare and exceptional. To our knowledge, this is the first report of a patient with second recurrence long after the first recurrence.
Late recurrence of breast cancer is considered to result from micrometastases or solitary dormant cancer cells that are reactivated and subsequently proliferate [15–17]. Tumor cells that have reached distant organs but have not yet formed clinically detectable metastatic lesions are referred to as disseminated tumor cells (DTCs) [18]. In patients, most detected DTCs exist as quiescent single cells [19]. Among the various organs in which DTCs can be found, particular attention has been paid to the bone marrow, as DTCs in this compartment are largely non-proliferative and can persist for prolonged periods [20]. Since the preoperative detection of DTCs in the bone marrow has been shown to be an independent prognostic marker of late recurrence, investigation of bone marrow DTCs may, in the future, become a standard test in patients with breast cancer [21]. Clear evidence regarding the optimal frequency of postoperative surveillance for breast cancer remains limited. This case suggests that maintaining long-term surveillance, for as long as is feasible, may be beneficial for the early detection of recurrence.
Multivariate analyses have identified ER and PgR positivity as factors associated with the timing of postoperative recurrence, with positive cases showing a significantly greater tendency toward late recurrence [22]. Furthermore, late recurrence has been reported to be more responsive to treatment and to have a better prognosis than early recurrence [23,24]. In the present case, however, approximately 5 months had elapsed from the time the patient first noticed lymphadenopathy and bilateral pleural effusion to the time she was referred to our hospital, resulting in a delayed diagnosis and the inability to initiate treatment.
In conclusion, we experienced an extremely rare case of breast cancer that developed a second recurrence 36 years after the initial surgery and 18 years after the first recurrence. This case underscores the need to remain vigilant for recurrence at any time in patients with a history of breast cancer and suggests that recurrence should be considered when new symptoms emerge.
Sources
1. Bray F, Laversanne M, Sung H et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 2024; 74 (3): 229–263. doi: 10.3322/caac.21834.
2. Robertson C, Arcot Ragupathy SK, Boachie C et al. The clinical effectiveness and cost-effectiveness of different surveillance mammography regimens after the treatment for primary breast cancer: systematic reviews registry database analyses and economic evaluation. Health Technol Assess 2011; 15 (34): v–vi, 1–322. doi: 10.3310/hta15340.
3. Drukteinis JS, Gombos EC, Raza S et al. MR imaging assessment of the breast after breast conservation therapy: distinguishing benign from malignant lesions. Radiographics 2012; 32 (1): 219–234. doi: 10.1148/rg.321115016.
4. Anderson SJ, Wapnir I, Dignam JJ et al. Prognosis after ipsilateral breast tumor recurrence and locoregional recurrences in patients treated by breast-conserving therapy in five national surgical adjuvant breast and bowel project protocols of node-negative breast cancer. J Clin Oncol 2009; 27 (15): 2466–2473. doi: 10.1200/JCO.2008.19.8424.
5. Komoike Y, Akiyama F, Iino Y et al. Ipsilateral breast tumor recurrence (IBTR) after breast-conserving treatment for early breast cancer: risk factors and impact on distant metastases. Cancer 2006; 106 (1): 35–41. doi: 10.1002/cncr.21551.
6. Saphner T, Tormey DC, Gray R. Annual hazard rates of recurrence for breast cancer after primary therapy. J Clin Oncol 1996; 14 (10): 2738–2746. doi: 10.1200/JCO. 1996.14.10.2738.
7. Takada K, Kashiwagi S, Kawajiri H et al. Late recurrence of breast cancer 32 years after surgery – a case report. [Japanese]. Gan To Kagaku Ryoho 2004; 31 (4): 678.
8. Yin D, Zhang G, Zhao L et al. Pulmonary resection and systemic lymph node dissection in a patient with breast cancer WHO had a 33-year disease-free interval. World J Surg Oncol 2015; 13 : 150. doi: 10.1186/s12957-015-0565-y.
9. Okutani D, Andou A, Yamadori I et al. Pulmonary metastasis from breast cancer 38 years after mastectomy; report of a case. [Japanese]. Kyobu Geka 2014; 67 (7): 592-594.
10. Tashima Y, Kawano K. A case of local recurrence developing thirty-nine years after mastectomy for breast cancer. [Japanese]. Gan to Kagaku Ryoho 2014; 41 (3): 357-9.
11. Oshima K, Kikumori K, Yanagawa T et al. A case of late recurrence of breast cancer with chest wall recurrence 43 years after surgery. [Japanese]. Gan To Kagaku Ryoho 2021; 48 (13): 1846–1848.
12. Pagani O, Senkus E, Wood W et al. International guidelines for management of metastatic breast cancer: can metastatic breast cancer be cured? J Natl Cancer Inst 2010; 102 (7): 456–463. doi: 10.1093/jnci/djq029.
13. Lim GH, Alcantara VS, Ng RP et al. Patterns of breast cancer second recurrences in patients after mastectomy. Breast Cancer Res Treat 2022; 196 (3): 583–589. doi: 10.1007/s10549-022-06772-4.
14. Wapnir IL, Gelber S, Anderson SJ et al. Poor prognosis after second locoregional recurrences in the CALOR trial. Ann Surg Oncol 2017; 24 (2): 398–406. doi: 10.1245/s10434-016-5571-y.
15. Pedersen RN, Mellemkjær L, Ejlertsen B et al. Mortality after late breast cancer recurrence in Denmark. J Clin Oncol 2022; 40 (13): 1450–1463. doi: 10.1200/JCO.21.02062.
16. Giannakeas V, Narod SA. A generalizable relationship between mortality and time-to-death among breast cancer patients can be explained by tumour dormancy. Breast Cancer Res Treat 2019; 177 (3): 691–703. doi: 10.1007/s10549-019-05334-5.
17. Chen X, Fan Y, Xu B. Distinct characteristics and metastatic behaviors of late recurrence in patients with hormone receptor-positive/human epidermal growth factor receptor 2-negative breast cancer: a single institute experience of more than 10 years. Clin Breast Cancer 2018; 18 (6): e1353–e1360. doi: 10.1016/j.clbc.2018.07.014.
18. Kang Y, Pantel K. Tumor cell dissemination: emerging biological insights from animal models and cancer patients. Cancer Cell 2013; 23 (5): 573–581. doi: 10.1016/j.ccr.2013.04.017.
19. Naumov GN, MacDonald IC, Weinmeister PM et al. Persistence of solitary mammary carcinoma cells in a secondary site: a possible contributor to dormancy. Cancer Res 2002; 62 (7): 2162–2168.
20. Ghajar CM, Peinado H, Mori H et al. The perivascular niche regulates breast tumour dormancy. Nat Cell Biol 2013; 15 (7): 807–817. doi: 10.1038/ncb2767.
21. Tjensvoll K, Nordgård O, Skjæveland M et al. Detection of disseminated tumor cells in bone marrow predict late recurrences in operable breast cancer patients. BMC Cancer 2019; 19 (1): 1131. doi: 10.1186/s12885-019-6268-y.
22. Takeuchi H, Tsuji K, Ueo H. Prediction of early and late recurrence in patients with breast carcinoma. Breast Cancer 2005; 12 (3): 161–165. doi: 10.2325/jbcs.12.161.
23. Courdi A, Largillier R, Ferrero JM et al. Early versus late local recurrences after conservative treatment of breast carcinoma: differences in primary tumor characteristics and patient outcome. Oncology 2006; 71 (5–6): 361–368. doi: 10.1159/000107771.
24. van der Sangen MJC, van de Poll-Franse LV, Roumen RMH et al. The prognosis of patients with local recurrence more than five years after breast conservation therapy for invasive breast carcinoma. Eur J Surg Oncol 2006; 32 (1): 34–38. doi: 10.1016/j.ejso.2005.10.005.
Labels
Paediatric clinical oncology Surgery Clinical oncologyArticle was published in
Clinical Oncology

2026 Issue 2
- Safety and Tolerance of Metamizole in Postoperative Analgesia in Children
- Metamizole in perioperative treatment in children under 14 years – results of a questionnaire survey from practice
- Obstacle Called Vasospasm: Which Solution Is Most Effective in Microsurgery and How to Pharmacologically Assist It?
- Possibilities of Using Metamizole in the Treatment of Acute Primary Headaches
- Current Insights into the Antispasmodic and Analgesic Effects of Metamizole on the Gastrointestinal Tract
-
All articles in this issue
- Není ct jako CT
- Treatment of Castleman disease from the perspective of the year 2026
- Ewing sarcoma – current diagnostic and therapeutic approaches
- Circulating tumor DNA as a biomarker for the prediction of minimal residual disease and the individualization of adjuvant therapy in colorectal cancer – a review of current evidence and perspectives
- Intravascular propagation of adenomyosis within the blood vessels of the myometrium and uterine leiomyoma in a patient harboring germline mutation in the fumarate hydratase gene – a rare finding simulating malignancy
- Second recurrence of breast cancer 36 years after initial surgery and 18 years after the first recurrence – an extremely rare case
- FDG-PET/CT: metabolically active solitary lesion in the gluteal region subcutaneously in a patient with breast cancer – granuloma or metastasis?
- Cancer patients admitted to intensive care unit
- Nádory GIT – chemoterapie a precizní léčba se synergicky doplňují
- Snižování škod rizikového chování v onkologické prevenci
- AKTUALITY Z NÁRODNÍHO ÚSTAVU PRO VÝZKUM RAKOVINY
- Clinical Oncology
- Journal archive
- Current issue
- About the journal
Most read in this issue
- Treatment of Castleman disease from the perspective of the year 2026
- FDG-PET/CT: metabolically active solitary lesion in the gluteal region subcutaneously in a patient with breast cancer – granuloma or metastasis?
- Ewing sarcoma – current diagnostic and therapeutic approaches
- Circulating tumor DNA as a biomarker for the prediction of minimal residual disease and the individualization of adjuvant therapy in colorectal cancer – a review of current evidence and perspectives
