Surveillance of invasive pneumococcal disease and serotypes coverage of pneumococcal vaccines in the Czech Republic between 2007 and 2024
Authors:
J. Kozáková 1
; S. Vohrnová 1,2
; P. Křížová 1
; M. Malý 3
Authors‘ workplace:
National Reference Laboratory for Streptococcal Infections, Centre for Epidemiology and Microbiology, National Institute of Public Health, Prague, Czech Republic
1; 3rd Faculty of Medicine, Charles University, Prague, Czech Republic
2; Unit of Biostatistics, National Institute of Public Health, Prague, Czech Republic
3
Published in:
Epidemiol. Mikrobiol. Imunol. 75, 2026, č. 1, s. 24-37
Category:
Original Papers
doi:
https://doi.org/10.61568/emi/11-6659/20260202/142617
Overview
Objective: This study presents the epidemiological situation of invasive pneumococcal disease (IPD) in the Czech Republic between 2007 and 2024.
Methods: The source of data for the study was the nationwide IPD surveillance programme, which started in 2008, 2007 being the pilot year of the programme. Surveillance data on the IPD were obtained by combining data from the National Reference Laboratory for Streptococcal Infections (NRL) and data from EpiDat and the Infectious Disease Information System (ISIN), respectively, always excluding duplicate records.
Results: Between 2007 and 2024, the total of 7,144 cases of IPD were recorded in the Czech Republic, with a total incidence of 3.7/100,000 population, varying in the individual years between 2.3 and 5.6/100,000. The age-specific average annual incidence over the entire period was at the following levels: children under one year – 6.1/100,000, children 1–4 years – 3.9/100,000, adults 40–64 years – 3.7/100,000, adults 65 years and older – 9.6/100,000. For the period 2007 to 2024, serotypes 3, 19A, and 8 were the most common serotypes detected in IPDs. In the 0–4 years age group, serotypes 3, 19A, and 14 were the most common serotypes; in adult aged 65 years and older, serotypes 3, 19A, and 22F were the most common serotypes. Over the years, there was a decline of serotypes included in pneumococcal conjugate vaccines (PCVs), with the exception of serotypes 3 and 19A, and an increase of serotypes not included in pneumococcal vaccines. Following the introduction of vaccination against pneumococcal diseases in the Czech Republic, the occurrence of serotype 4 initially declined; however, in recent years, its occurrence has increased. The percentage coverage of serotypes causing IPD with currently recommended pneumococcal vaccines in 2024 was as follows: PCV13 – 42.8%, PCV15 – 48.5%, PCV20 – 60.7%, PCV21 – 62.8%, PPV23 – 66.9%. The percentage coverage of serotypes in the Czech Republic by current vaccines is comparable to that in other countries; however, in the case of potential new vaccines, it is lower than reported in other countries.
Conclusion: Between 2007 and 2024, there was a decrease in IPD morbidity in the paediatric population and an increase in IPD morbidity in adults aged 65 years and older. The data presented may serve as a basis for updating recommendations for vaccination against pneumococcal diseases by the Czech Vaccination Society. Given the serotype coverage of the individual vaccines, we recommend the use of higher-valent conjugate vaccines PCV15 or PCV20 in children, rather than PCV13. The development of pneumococcal vaccines is based on data from IPD surveillance programmes in countries other than the Czech Republic; therefore, the serotype composition of these vaccines may not fully correspond to the serotypes circulating in the Czech Republic. The development of new pneumococcal vaccines is ongoing. It is essential to continue detailed surveillance of IPD in order to update and optimize vaccination strategy against pneumococcal diseases in the Czech Republic.
Keywords:
vaccination – Streptococcus pneumoniae – invasive pneumococcal disease – serotyping
INTRODUCTION
Streptococcus pneumoniae (pneumococcus) is a Gram-positive, catalase-negative, facultative anaerobic bacterium. Commonly found in the upper respiratory tract of healthy individuals, it is detected in 30 to 70% of young children and up to 5% of adults, more commonly in adults living in households with young children [1]. S. pneumoniae can cause a wide range of diseases from upper respiratory tract infections such as rhinosinusitis, pharyngitis, otitis media, to uncomplicated pneumonia, to pneumonia with a severe course, with destruction of lung tissue, pyothorax and sepsis, septic arthritis, sepsis and meningitis. Severe infections caused by S. pneumoniae are globally referred to as invasive pneumococcal disease (IPD).
Young children, older adults, and all age groups of the population with immunocompromising conditions are most at risk for a severe course of pneumococcal disease. Vaccination with pneumococcal vaccines is recommended for the above population groups. Currently, the following pneumococcal conjugate vaccines (PCVs) are available in the Czech Republic: 13 - valent PCV13 contains serotypes 1, 4, 5, 6B, 7F, 9V, 14, 18C, 19F, 23F, 3, 6A, 19A; 15 - valent PCV15 contains serotypes 1, 4, 5, 6B, 7F, 9V, 14, 18C, 19F, 23F, 3, 6A, 19A, 22F, 33F; 20-valent PCV20 contains serotypes 1, 4, 5, 6B, 7F, 9V, 14, 18C, 19F, 23F, 3, 6A, 19A, 22F, 33F, 8, 10A, 11A, 12F, 15B. In addition, a 23-valent pneumococcal polysaccharide vaccine 23 (PPV23) is available containing serotypes 1, 2, 3, 4, 5, 6B, 7F, 8, 9N, 9V, 10A, 11A, 12F, 14, 15B, 17F, 18C, 19A, 19F, 20, 22F, 23F, 33F. All of the above conjugated vaccines are currently approved for use in the paediatric population in the Czech Republic [2]. In the adult population, the recommended vaccine is PCV15 in combination with PPV23 or PCV20 alone [3].
The development of new pneumococcal vaccines is ongoing. A 21-valent PCV21 vaccine was approved in the USA in June 2024 for the adult population and contains serotypes 3, 6A, 7F, 8, 9N, 10A, 11A, 12F, 15A, 15C, 16F, 17F, 19A, 20A, 22F, 23A, 23B, 24F, 31, 33F, 35B [4].
Two 24-valent PCV24 vaccines containing serotypes 1, 2, 3, 4, 5, 6A, 6B, 7F, 8, 9N, 9V, 10A, 11A, 12F, 14, 15B, 17F, 18C, 19A, 19F, 20B, 22F, 23F, 33F are under development [5, 6]. A 25-valent PCV25 vaccine containing serotypes 1, 2, 3, 4, 5, 6B, 6C, 7F, 8, 9N, 9V, 10A, 12F, 14, 15A, 15B, 16F, 18C, 19A, 19F, 22F, 23F, 24F, 33F, 35B, and 31-valent PCV31 vaccine containing serotypes 1, 2, 3, 4, 5, 6A, 6B, 7C, 7F, 8, 9N, 9V, 10A, 11A, 12F, 14, 15A, 15B, 16F, 17F, 18C, 19A, 19F, 20B, 22F, 23A, 23B, 23F, 31, 33F, 35B are in the second stages of testing [7, 8, 9].
In the Czech Republic, vaccination against pneumococcal diseases was initiated with the PPV23 vaccine, which was recommended for patients with immunocompromising conditions and for the elderly aged 65 years and older; the vaccine was not covered by health insurance. The 7-valent PCV7 vaccine containing serotypes 4, 6B, 9V, 14, 18C, 19F, 23F was registered and used in the Czech Republic from 2005 and was intended for children under 2 years of age and was not covered by public health insurance [10]. In 2010, vaccination with PCV10 and PCV13 vaccines was introduced in the Czech Republic for children aged 2 months to 5 years, of which PCV10 was fully covered by public health insurance and PCV13 was covered up to the price of PCV10. From 2016, reimbursed vaccination with PCV13 was introduced for patients with specified diagnoses and from 2017 for the elderly age group aged 65 years and older. In 2022, PCV15 and PCV20 vaccines were recommended for seniors aged 65 years and older and for patients with immunocompromising conditions, and PCV15 for children aged 2 months and older. In April 2024, PCV20 was also approved for use in children 2 months of age and older. Detailed information on the vaccination schedules for each population group is available on the website of the Czech Vaccination Society [11]. Vaccination against pneumococcal diseases is not compulsory in the Czech Republic but is recommended. The proportion of children born in 2022 vaccinated with at least one dose of PCV was 76.4%, and the proportion of seniors over 65 years of age vaccinated against pneumococcal diseases with any vaccine was 22.5% at the end of 2023 [12].
Along with the introduction of PCV in the Czech Republic, a study was conducted to monitor the occurrence of IPD between 1997 and 2006 and to analyse the coverage of the serotypes with the vaccines used at that time [13]. The year 2007 was a pilot year of the nationwide IPD surveillance programme in the Czech Republic. The year 2008 was the first year when the IPD surveillance programme was conducted according to the Methodological Guideline for IPD Surveillance (Methodological Guideline – Invasive Pneumococcal Disease Epidemiological Vigilance System) [14]. The IPD surveillance programme was anchored in the Czech legislation, sending samples of S. pneumoniae isolated from primary sterile material to the National Reference Laboratory for Streptococcal Infections (NRL) became mandatory for bacteriological laboratories, as well as the obligation for regional public health authorities to record IPD in the EpiDat database, which operated until 2017. Since 2018, cases have been recorded in the database of the Infectious Disease Information System (ISIN). IPD surveillance data are obtained by merging data from the NRL database and the EpiDat/ISIN database, always excluding duplicate records. IPD cases that are recorded only in the NRL database are reported back to the ISIN database. The IPD surveillance database includes cases that meet both the European and Czech case definitions of IPD. This definition was set out in the Methodological Guideline on Surveillance IPD and in Decree No. 275/2010 Coll., Annex 21, which was replaced by Decree No. 389/2023, Annex 21 as of 1 January 2024 [14, 15, 16].
At the NRL, all isolates received are checked for correct identification of S. pneumoniae and serotyped. Until 2012, serotyping was performed using the Quellung (Neufeld) reaction, since 2013, serotyping is performed using a combination of the polymerase chain reaction (PCR) with the Quellung reaction [17].
MATERIAL AND METHODS
IPD case definition
A case of IPD is defined as a severe disease with laboratory confirmed presence of S. pneumoniae in a clinical specimen obtained from a normally sterile site. This definition is given by Decree No 389/2023, Annex 21 and the IPD Surveillance Methodological Guide and is identical to the European IPD definition [14, 15, 16].
Data sources
Epidemiological data for the study were obtained from the nationwide IPD surveillance programme. Only completely closed datasets from the years 2007–2024 were included in the study. Isolates from IPD are sent to the NRL by microbiological laboratories in the Czech Republic.
ISIN and EpiDat records of IPD cases and S. pneumoniae isolates were obtained in accordance with Czech legislation. Ethics committee approval was not required as no personal data were included in the study.
Identification and serotyping in the NRL
All S. pneumoniae isolates received at the NRL were checked for correct identification by optochin susceptibility testing and solubility testing in 10% sodium deoxycholate solution (bile solubility test). Since 2013, all S. pneumoniae isolates received at the NRL have been tested for the presence of the cpsA gene as an additional method of verification of identification. In case of unclear results, other identification methods such as latex agglutination, reaction with pneumococcal OMNI serum, biochemical identification, microscopy, catalase test or molecular methods (PCR, MLST) were used. Serotyping of S. pneumoniae was performed until 2012 using the Quellung reaction. In 2013, a serotyping method combining end-point multiplex PCR and Quellung reaction was introduced [17]. In 2014, the real-time PCR method was used for identification and typing from clinical specimens [18].
PCR reactions include multiple primers for a single serotype, serogroup, or combinations of serotypes and serogroups. The Quellung reaction then enables the identification of the serotype from pure cultures of isolates. It is not possible to perform the Quellung reaction on clinical specimens. The Quellung reaction can be observed under a phase-contrast microscope and requires experienced personnel to perform the test. The Quellung reaction cannot reliably distinguish between some serotypes (e.g 20A,20B, and 20C); whole-genome sequencing (WGS), which is not part of routine testing in the NRL, is appropriate method for accurate differentiation. Serotypes 20A, 20B, and 20C are included in the analyses as serogroup 20. The determination of serotypes 15B and 15C is performed at the NRL using the Quellung reaction, although WGS is a more reliable technique.
S. pneumoniae isolates with undetermined serotypes occured when the IPD case was recorded in the EpiDat/ ISIN database but the S. pneumoniae isolate was either not delivered to the NRL for serotyping or could not be successfully re-cultivated after delivery. These situations occurred randomly, without any selection bias.
Serotype coverage of pneumococcal vaccines
By comparing the occurrence of each serotype that caused IPD with the serotypes contained in previous, current and possible future vaccines PCV7, PCV10, PCV13, PCV15, PCV20, PCV21, PPV23, PCV24, PCV25 and PCV31, the percentage coverage of serotypes by each pneumococcal vaccine was determined. The proportion of each serotype was determined relative to the total number of IPD cases in each year. In PCV21, serotype 20A is counted as serotype 20.
The data were processed using statistical software Stata, release 17 (StataCorp LLC, College Station, TX, U.S.A.).
RESULTS
Epidemiological situation of IPD
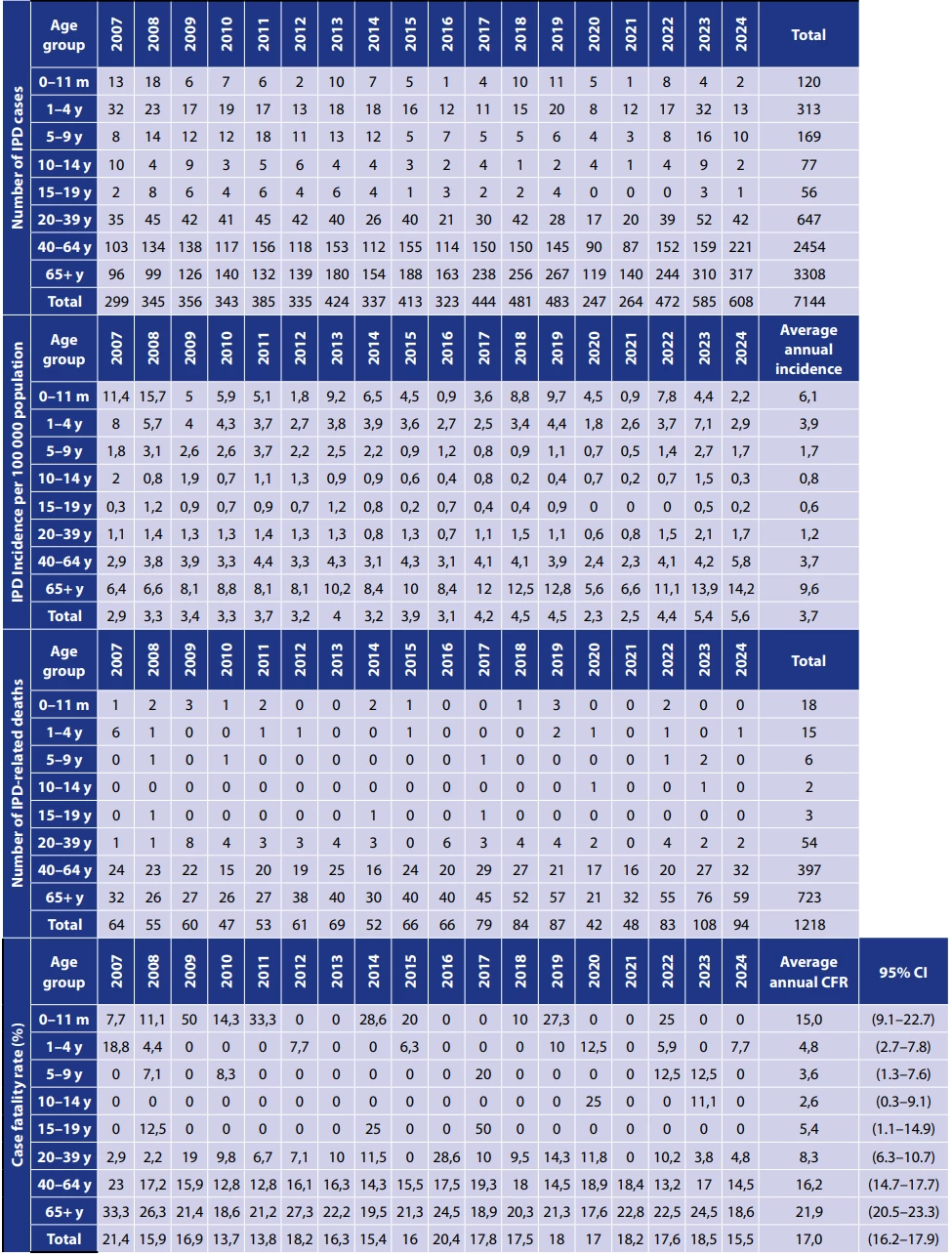
Between 2007 and 2024, a total of 7,144 cases of IPD were recorded, with a total incidence of 3.7/100,000 population, ranging between 2.3 and 5.6 cases per 100,000 across individual years. The lowest number of IPD was in 2020, when 247 IPD cases were recorded, and the incidence was at 2.3/100,000. The highest number of IPD was in 2024 when 608 IPD cases were recorded and the incidence was 5.6/100,000 – Table 1, Figure 1.
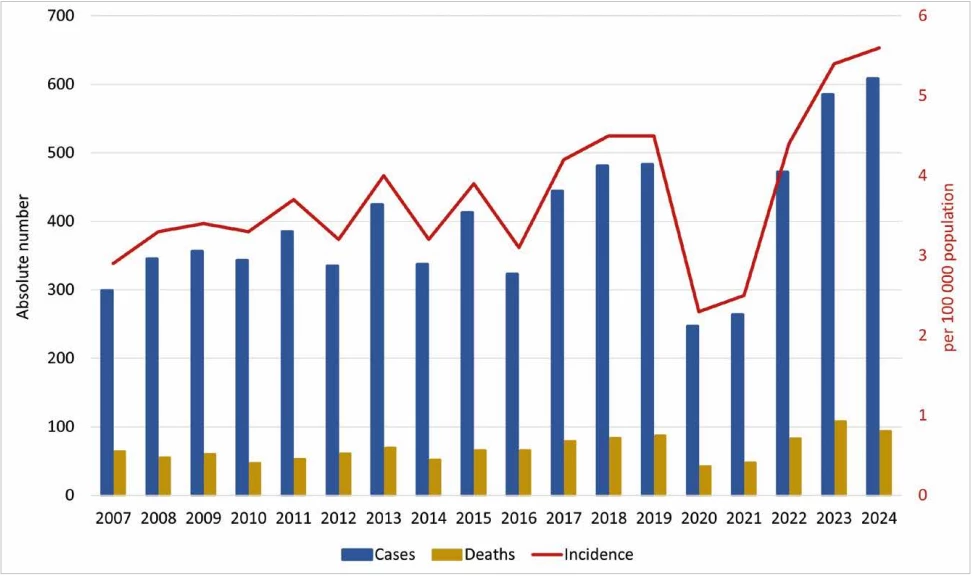
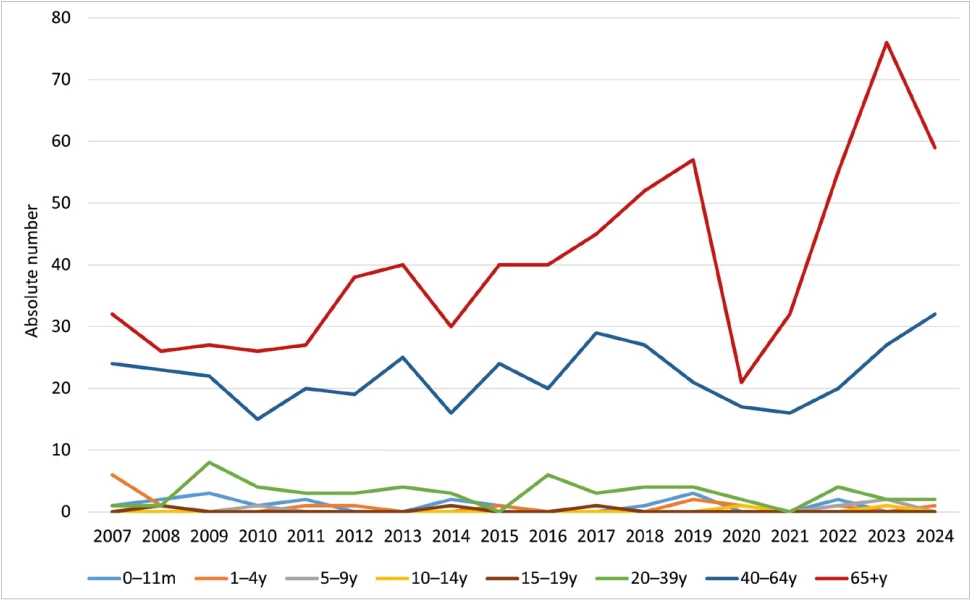
Age-specific average annual incidence was highest in the 65 years and older age group, with 9.6/100,000; the lowest was recorded in 2020 at 5.6/100,000 (119 IPD cases), and the highest in 2024 at 14.2/100,000 (310 IPD cases). For the age group of children under 1 year, the average incidence was 6.1/100,000, the lowest was recorded in 2016 and 2021 – 0.9/100,000 (1 IPD case), the highest in 2008 – 15.7/100,000 (18 IPD cases). For children in the age group 1–4 years, the average age-specific incidence was 3.9/100,000, the lowest was recorded in 2020 – 1.8/100,000 (8 IPD cases), the highest in 2007 – 8.0/100,000 (32 IPD cases) – Table 1, Figure 2.
The proportions are supplemented with 95% confidence interval.
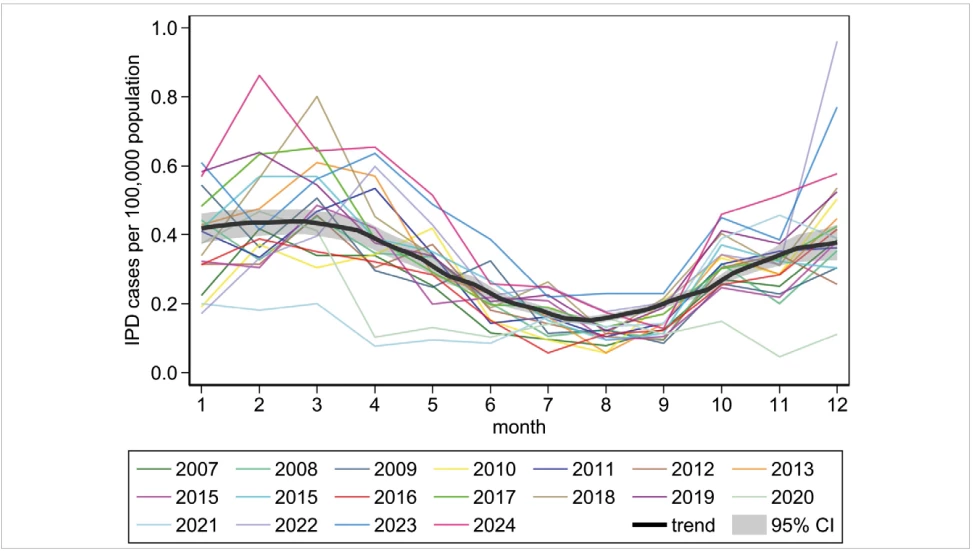
The IPD incidence shows a seasonal pattern, coinciding with the circulation of respiratory viruses in the population. The highest IPD incidence was repeatedly recorded from January to April, followed by a gradual decline during the year, with the lowest incidence in August, followed by a gradual increase in the autumn – Figure 3. In 2020, the IPD season began in line with typical seasonal pattern, but there was not the expected increase in IPD in the autumn. In 2021, there was a very low IPD incidence in the months of January to March, and then in the autumn of 2021, there was again an increase in IPD incidence, similar to the patterns observed between 2007 and 2019.
Between 2007 and 2024, a total of 1218 deaths related to IPD were recorded, with an average of 68 deaths per year – Table 1, Figure 4. The fewest deaths related to IPD were recorded in 2020 – 42 deaths, the most occurred in 2023 – 108 deaths – Figure 1. In the age group of children under 5 years, an average of 2 deaths per year was recorded, whereas in the age group of adults aged 65 years and older, an average of 40 deaths per year was recorded. The total case fatality rate was 17.0% over the period, the lowest case fatality rate was in 2010 – 13.7%, the highest in 2007 – 21.4% – Table 1.
Serotypes causing IPD
Between 2007 and 2024, out of a total of 7144 IPD cases, the serotype (serotype/serogroup) was determined in 6359 IPD cases (89%), while in the remaining IPD cases do not have serotype determined. A total of 75 different serotypes and serogroups were identified over the period, and 6353 isolates were identified down to the serotype.
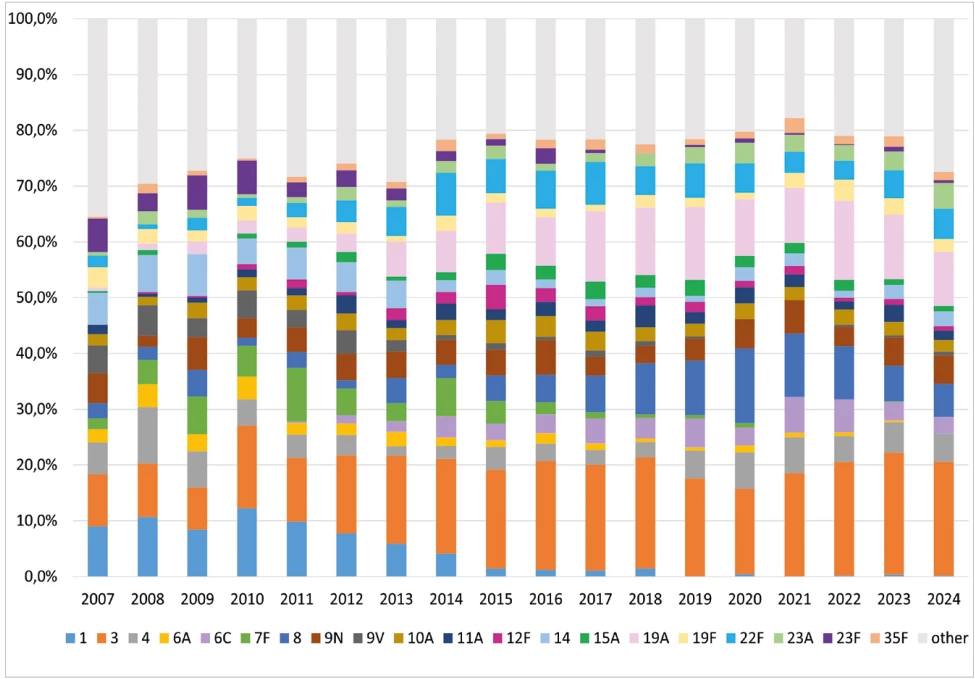
IPD were most frequently caused by serotype 3 (16.7%), 19A (8.2%), 8 (5.8%) and 4 and 22F (both 4.6%) – Table 2, Figure 5. Serotypes 3 (10.0%), 19A (9.2%), and 19F (7.5%) were the most common serotypes recorded in the age group of children under 1 year; serotypes 3 (16.6%), 19A (11.2%), and 14 (8.6%) were the most common serotypes in children aged 1 to 4 years. In the age group 65 years and older, serotypes 3 (18.9%), 19A (8.6%), and 22F (5.8%) were predominant.
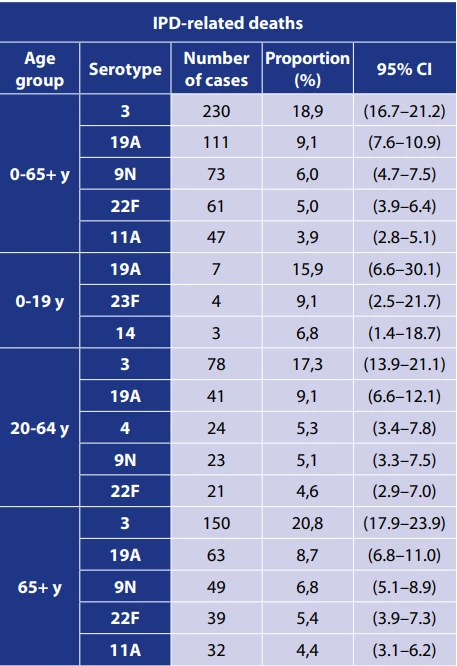
Between 2007 and 2024, of the 1218 IPD-related deaths, 1121 cases (92%) were serotyped, and 59 serotypes and serogroups were identified, with up to 1119 cases identified down to the serotype. For deaths related to IPD, serotype 3 (18.9%), 19A (9.1%), 9N (6.0%), and 22F (5.0%) were the most frequently identified serotypes – Table 3. In children aged 0 to 19 years, serotypes 19A (15.9%), 23F (9.1%), and 14 (6.8%) were the most frequently detected serotypes in IPD-related deaths. In the age group 65 years and older, serotypes 3 (20.8%), 19A (8.7%) and 9N (6.8%) were the predominant serotypes in IPD-related deaths.
The proportions are supplemented with 95% confidence interval (CI).
Case fatality rate caused by serotypes of S. pneumoniae
Among the 20 most frequent serotypes that caused IPD in all age groups in the Czech Republic between 2007 and 2024, the serotypes with the highest case fatality rate were 11A (31,5%), 6A and 12F – Table 4. Among the 10 most frequent serotypes in children under 5 years and adults 65 years and older, the highest case fatality rates were observed for serotypes 23F and 11A, respectively – Table 4.
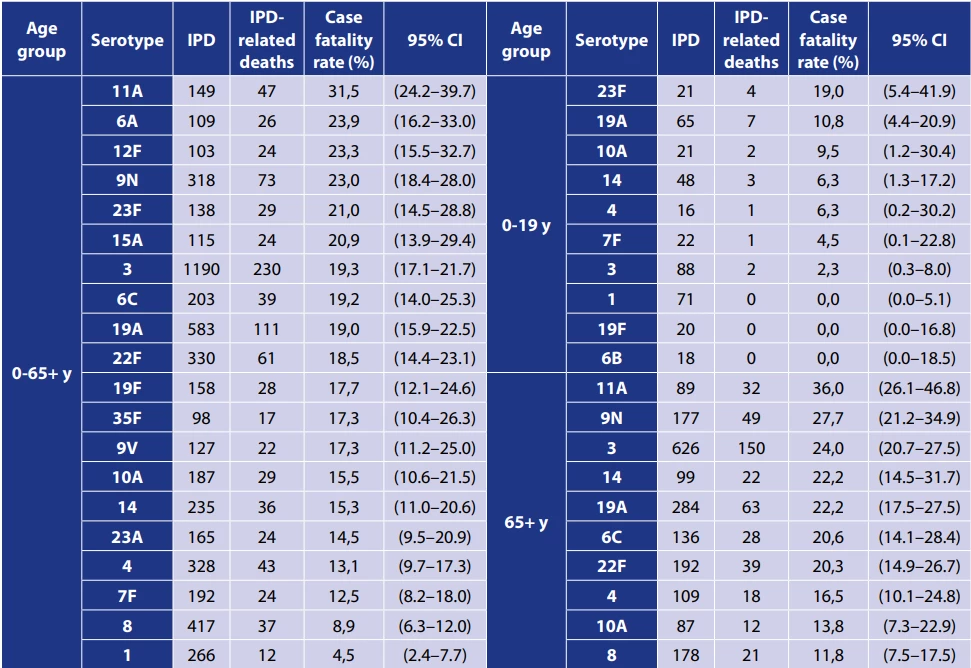
The case fatality rates are supplemented with 95% confidence interval (CI).
Pneumococcal vaccine coverage
The percentage vaccine coverage of serotypes that caused IPD in selected age groups between 2007 and 2024 in the Czech Republic was determined for vaccines PCV7, PCV10, PCV13, PCV15, PCV20, PCV21, PPV23, PCV24, PCV25 and PCV31 – Figure 6, 7, 8. In 2024, the percentage coverage of serotypes causing IPD overall, in the age groups of children under 5 years and in adults 65 years and older is shown in Table 5, in all age groups for current vaccines was the coverage as follows: PCV13 – 42.8%, PCV15 – 48.5%, PCV20 – 60.7%, PCV21 – 62.8%, PPV23 – 66.9%.
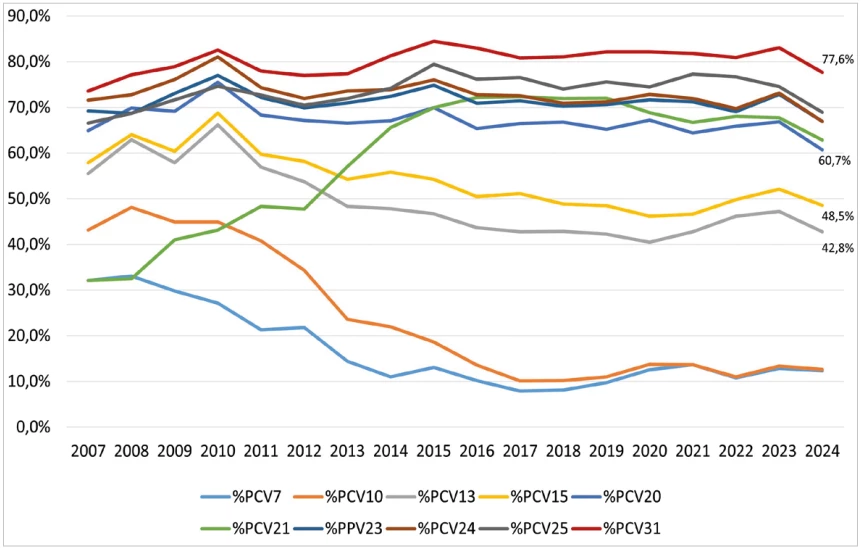
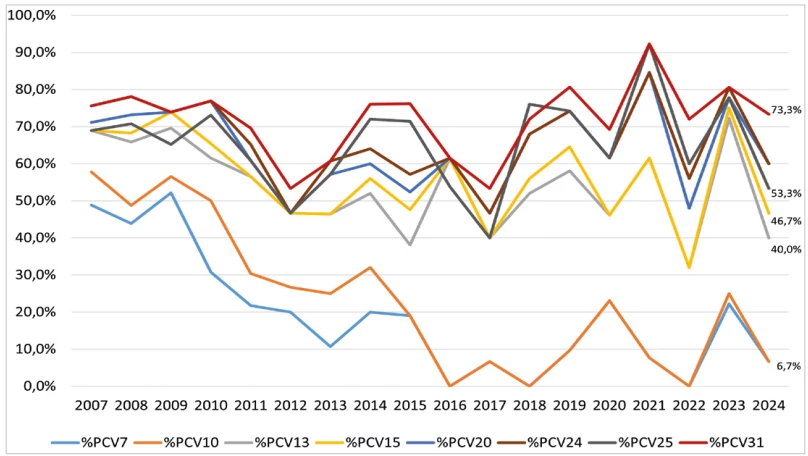
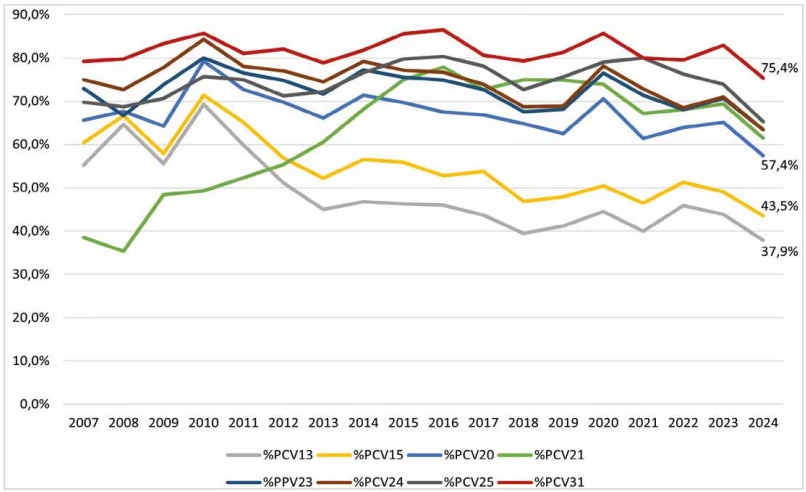
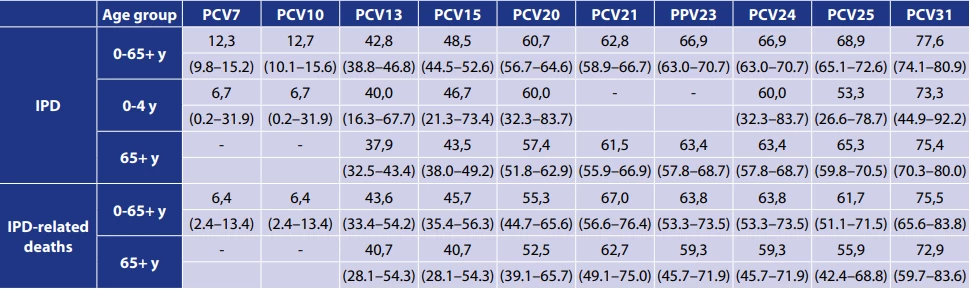
The percentages are supplemented with 95% confidence intervals (CI).
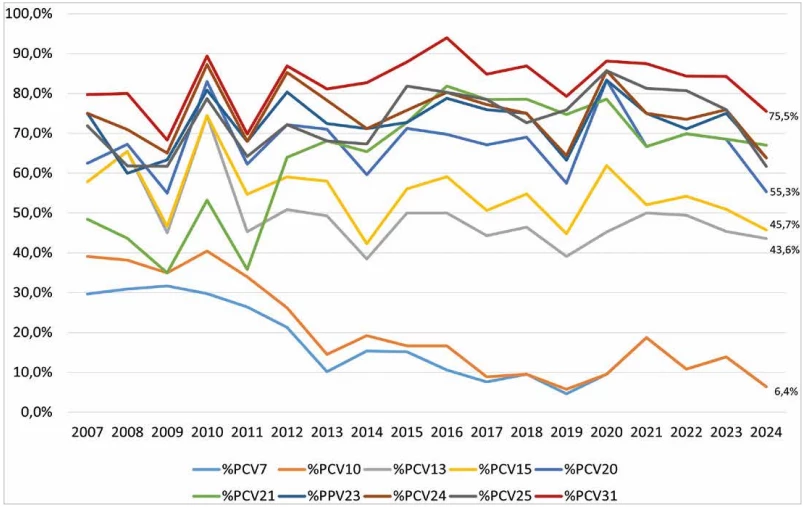
Between 2007 and 2024, a substantial decline of serotypes included in PCV7 and PCV10 was observed in all age groups. Serotypes covered by PCV13 and PCV15 showed a gradual decrease, while those included in PCV20, PPV23, PCV24, and PCV25 exhibited relatively stable occurrence. Over the period, a significant increase of serotypes included in PCV21 was noted, with a slight rise also observed for serotypes contained in PCV31. The increasing trend in PCV21 serotypes was particularly evident among adults aged 65 years and older
In children aged 0–4 years, the serotypes included in the respective vaccines fluctuated markedly over the period. Nevertheless, a tendency toward increasing occurrence was apparent for serotypes in PCV24, PCV25, and PCV31. Serotypes included in PCV13, PCV15, and PCV20 showed relatively stable occurrence, whereas a notable decline was observed in serotypes covered by PCV7 and PCV10.
The percentage of vaccine coverage for serotypes in IPD-related deaths in 2024 was determined for all previous, current, and potential future PCVs, both for all IPD-related deaths and specifically for IPD-related deaths in the age group of 65 years and older – Table 5. The coverage of current PCVs across all age groups in 2024 was as follows: PCV13 – 43.6%, PCV15 – 45.7%, PCV20 – 55.3%, PCV21 – 67.0%, PCV23 – 63.8%. The percentage vaccine coverage of serotypes causing IPD-related deaths between years 2007 and 2024 in the Czech Republic shows decline of serotype in PCV7 and PCV10, rise of serotypes in PCV21, and relatively stable occurrence of serotypes in the rest of the PCVs – Figure 9.
DISCUSSION
The data presented here show trends in the incidence of IPD and the frequency of different serotypes in the Czech Republic from 2007 to 2024. The use of vaccines against pneumococcal diseases has significantly reduced the incidence of IPD caused by serotypes included in the vaccines [19, 20, 21, 22]. After the introduction of PCV7, the occurrence of serotypes included in this vaccine decreased significantly in the Czech Republic [23]. In the years following the introduction of PCV10 and PCV13, further changes in the prevalence of IPD and selected serotypes occurred. The impact of vaccination on the decline of IPD in children in the Czech Republic between 2007 and 2017 has previously been demonstrated [24]. The findings from our study show a gradual decline in the incidence of IPD in the age groups of children up to 19 years, stagnation in the incidence of IPD in the age groups of 20 to 39 years and 40 to 64 years and, in contrast, an increase in the incidence of IPD in adults aged 65 years and older. Serotypes included in PCVs have been on the decline and, conversely, serotypes not included in PCVs have begun to occur at a higher rate (serotype replacement), which has been repeatedly described in other countries [19, 25, 26]. In 2020 and 2021, there was a decline in the incidence of IPD in all age groups in the Czech Republic. This trend was consistent with the global pattern of IPD incidence [27, 28]. The reason for the decline in IPD incidence can be attributed to the anti-epidemic measures in place to prevent the spread of COVID-19 infection.
In the post-COVID-19 pandemic years, a trend towards an increase in the incidence of IPD was observed in all age groups, to values higher than in the pre-pandemic period. In considering the causes of this situation, we can take into account the possible disruption of vaccination programmes during the pandemic years and further consider the possibility of an immune debt effect, whereby reduced social interaction during the pandemic years led to decreased exposure to common bacterial pathogens, potentially limiting natural immune stimulation [29, 30, 31].
Analysis of serotype occurrence shows that serotypes 3 and 19A are more prevalent in the Czech Republic despite the vaccination with PCV13. In a UK study of the efficacy of PCV7 and PCV13 vaccines, there was no significant efficacy of PCV13 against serotype 3 [32].
Other international studies have also described a lower efficacy of PCV13 against serotype 3, with an increasing incidence of IPD caused by serotype 3 even in countries with established vaccination with PCV13 [33, 34, 35, 36]. Studies indicate that PCV15 demonstrates superior immunogenicity (opsonophagocytic activity) against serotype 3 compared to other vaccines containing this serotype [37, 38]. Whether PCV15 will also provide improved clinical efficacy against serotype 3 needs to be confirmed through IPD surveillance and monitoring of population vaccination coverage. A UK publication described a stagnation in the incidence of serotype 19A in IPD [36].
In recent years, there has been a trend of increased incidence of some serotypes that are contained in the PCV10. Particularly in the adult population aged 40 years and above, there has been an increased incidence of cases caused by serotype 4. Serotype 4 is known for its potential to cause outbreaks, as has been repeatedly described [39, 40]. Further monitoring of the incidence of serotypes causing IPD in the Czech Republic is needed to assess whether this is a sustained trend or just a transient fluctuation.
When comparing the percentage coverage of circulating serotypes by the individual pneumococcal vaccines, a decrease in the prevalence of serotypes included in current vaccines (PCV13, PCV15, PCV20, PPV23) and potential future vaccines (PCV21, PCV24, PCV25 and PCV31) in 2024 can be observed. The serotype coverage by existing vaccines in the Czech Republic is comparable to other countries, but as far as possible new vaccines are considered, it is lower than reported in other countries. In the USA, the coverage of serotypes from 2018 to 2022 contained in the PCV21 vaccine is reported to be 85% for the age group 65 years and older and 54% for the PCV20 vaccine [41]. In Canada, in the population aged 65 years and older, the serotype coverage with PCV21 vaccine is 81.5% and with PCV20 vaccine 52.2% [42]. The development of pneumococcal vaccines is based on data from IPD surveillance programmes of countries other than the Czech Republic, therefore the serotype composition of vaccines may not fully correspond to the serotypes circulating in the Czech Republic.
The limitation of calculating percentage vaccine coverage of serotypes in the age group of children under 5 years is the low number of cases, which means that even a single case can markedly change the resulting percentage coverage of vaccines. For instance, the difference between PCV25 coverage (53.3%) and PCV20/ PCV24 coverage (60.0%) is due to one case of serotype 11A, which is not included in PCV25.
The data presented are from the national IPD surveillance programme, reporting of cases and sending samples to the NRL is mandatory by law. A potential limitation in comparing IPD incidence data from the Czech Republic with international studies is the relatively low number of blood cultures collected in the Czech Republic compared to other European countries. The Surveillance report Antimicrobial Resistance in the EU/EEA (EARS-Net) 2023 states that in the Czech Republic, the blood culture rate (blood culture sets per 1,000 patient –days) was 18.2, which is similar to Hungary, and the IPD incidence was also comparable [43, 44]. In contrast, Slovenia had a blood culture rate of 44.7, and its IPD incidence was more than twice as high as in the Czech Republic. It may have been the case that in the Czech Republic mainly clinically more severe cases were included in the IPD surveillance programme. In that case it can be assumed that IPD incidence would be even higher than presented in this study. It is important to note that direct comparison of IPD incidence between countries is also hindered by differences in pneumococcal vaccination programs [45].
CONCLUSION
The presented study shows that the incidence of IPD in the Czech Republic in children tends to decrease in the long term, but there is an increased incidence of IPD in the elderly population. The data presented may serve as a basis for updating recommendations for vaccination against pneumococcal diseases by the Czech Vaccination Society. According to the data, these recommendations should be focused on both children and the elderly population. Given the serotype coverage of the individual vaccines, we recommend the use of higher-valent conjugate vaccines PCV15 or PCV20 in children, rather than PCV13. Serotype 3 accounts for a substantial proportion of IPD cases in children; should PCV15 demonstrate superior clinical efficacy in protecting against serotype 3, we recommend prioritizing the use of this vaccine in children, also taking into account the PCV15 vaccination schedule. The COVID-19 pandemic has shown that the incidence of IPD is dynamic, and a potential future epidemics or pandemics may have a similar impact on IPD incidence. The development of pneumococcal vaccines is based on data from IPD surveillance programmes in countries other than the Czech Republic; therefore, the serotype composition of the vaccines may not fully correspond to the serotypes circulating in the Czech Republic. The development of new pneumococcal vaccines is ongoing. It is essential to continue detailed IPD surveillance to enable the updating of an appropriate vaccination strategy against pneumococcal diseases in the Czech Republic.
Acknowledgments We thank all microbiologists, epidemiologists, and clinical physicians for their cooperation in implementing the IPD surveillance programme in the Czech Republic. We also thank RNDr. Jitka Motlová, CSc., for providing the IPD surveillance data from 2007 to 2011. Project support Supported by the programme project of the Ministry of Health of the Czech Republic with reg. no. NU22-09-00433.
Sources
1. Greenberg D, Broides A, Blancovich I, et al. Relative importance of nasopharyngeal versus oropharyngeal sampling for isolation of Streptococcus pneumoniae and Haemophilus influenzae from healthy and sick individuals varies with age. J Clin Microbiol., 2004;42(10):4604–4609. doi: 10.1128/JCM.42.10.4604 - 4609.2004. PMID: 15472316; PMCID: PMC522367.
2. Recommendation of the Czech Vaccination Society. Available at: https://www.vakcinace.eu/doporuceni-a-stanoviska/doporuceni-ceske-vakcinologicke - spolecnosti-cls-jep-pro-ockovani-detia-adolescentu-ve-veku-0-17-let (Access: 22. 7. 2025).
3. Recommendation of the Czech Vaccination Society. Available at: https://www.vakcinace.eu/doporuceni-a-stanoviska/doporuceni-ceske-vakcinologicke - spolecnosti-cls-jep-pro-ockovani-dospelych-proti-po (Access: 22. 7. 2025).
4. Kobayashi M, Leidner AJ, Gierke R, et al. Use of 21-Valent Pneumococcal Conjugate Vaccine Among U.S. Adults: Recommendations of the Advisory Committee on Immunization Practices – United States, 2024. MMWR Morb Mortal Wkly Rep. 2024;73(36):793–798. DOI: 10.15585/mmwr.mm7336a3. PMID: 39264843.
5. Wassil J, Sisti M, Fairman J, et al. Evaluating the safety, tolerability, and immunogenicity of a 24-valent pneumococcal conjugate vaccine (VAX-24) in healthy adults aged 18 to 64 years: a phase 1/2, double-masked, dose-finding, active - controlled, randomised clinical trial. Lancet Infect Dis. 2024;24(3):308–318. doi: 10.1016/S1473-3099(23)00572-8. Epub 2023 Dec 4. PMID: 38061367.
6. Borys D, Rupp R, Smulders R, et al. Safety, tolerability and immunogenicity of a novel 24-valent pneumococcal vaccine in toddlers: A phase 1 randomized controlled trial. Vaccine, 2024;42(10):2560–2571. doi: 10.1016/j.vaccine.2024.02.001. Epub 2024 Feb 14. PMID: 38360475.
7. Inventprise. Preclinical results for a 25-valent pneumococcal conjugate vaccine using a novel linker platform technology. Available at: https://inventprise.com/2024/01/02/ inventprise-completes-vaccination-of-participants - in-a-phase2-dose-ranging-study-of-its-25-valent-pneumococcal-vaccinecandidate/ (Access: 22. 7. 2025).
8. Vaxcyte. Vaxcyte Reports Positive Data from Phase 2 Study of its 24-Valent Pneumococcal Conjugate Vaccine Candidate, VAX24, in Adults Aged 65 and Older and Full Six-Month Safety Data from Adult Phase 1/2 and Phase 2 Studies. 2023. Dostupné na: https://investors.vaxcyte.com/news-releases/news-release - details/vaxcyte-announces-positive-topline-results-vax-24-infantphase-2 (Access: 22. 7. 2025).
9. King LM, Lewnard JA. Health-economic burden attributable to novel serotypes in candidate 24 - and 31-valent pneumococcal conjugate vaccines. Vaccine, 2024;42(26):126310. DOI: 10.1016/j.vaccine.2024.126310. PMID: 39260055.
10. Chlíbek R. Bezpečnost konjugované 7valentní pneumokokové vakcíny. Pediatr. Praxi, 2009;10(1):26–30.
11. Recommendation of the Czech Vaccination Society. Available at: https://www.vakcinace.eu/doporuceni-a-stanoviska?tag=13 (Access: 22.7.2025).
12. ÚZIS. Aktuální data o proočkovanosti české populace. Available at: https://www.nzip.cz/data/ceo/analyticke-studie/ceo-vakcinace-prehled/ceo-vakcinace - prehled.pdf (Access: 22.7.2025).
13. Motlova J, Benes C, Kriz P. Incidence of invasive pneumococcal disease in the Czech Republic and serotype coverage by vaccines, 1997–2006. Epidemiol Infect, 2009;137(4):562–569. doi: 10.1017/S0950268808001301. Epub 2008 Sep 16. PMID: 18796171.
14. Methodological Guideline – Invasive Pneumococcal Disease Epidemiological Vigilance System. Bulletin of the Ministry of Health (2/2008). Available at: https://mzd.gov.cz/wpcontent/ uploads/wepub/1992/6150/V%C4%9Bstn%C3%ADk%20 02%202008.pdf (Access: 22.7.2025).
15. Decree No. 389/2023 Coll., Decree on the Epidemiological Surveillance System for Selected Infectious Diseases, amending Decree No. 473/2008 Coll. and Decree No. 275/2010 Coll. Annex No. 21: Epidemiological Surveillance System for Invasive Pneumococcal or Streptococcal Diseases. Available at: https://www.e - sbirka. cz/sb/2023/389/2024-11-15?zalozka=text (Access: 22.7.2025).
16. Case definitions for each infectious disease covered by EU surveillance, as published in the Official Journal of the European Union (Commission Implementing Decision (EU) 2018/945). Available at: https://eur-lex.europa.eu/legal - content/EN/TXT/ PDF/?uri=CELEX:32018D0945&from=EN#page=33 (Access: 22.7.2025).
17. Vacková Z, Klímová M, Kozáková J. A novel typing method and scheme for Streptococcus pneumoniae. Epidemiol Mikrobiol Imunol, 2013; 62 : 50–58. [Article in Czech].
18. Vacková Z, Lžičařová D, Stock NK, Kozáková J. Real-time PCR detection of Neisseria meningitidis, Haemophilus influenzae and Streptococcus pneumoniae DNA in clinical specimens. Epidemiol Mikrobiol Imunol., 2015;64(4):222–230. ISSN 1210-7913. [Article in Czech].
19. Waight PA, Andrews NJ, Ladhani S, et al. Effect of the 13-valent pneumococcal conjugate vaccine on invasive pneumococcal disease in England and Wales 4 years after its introduction: an observational cohort study. Lancet Infect Dis., 2015;15(5):535 – 543. DOI: 10.1016/S1473-3099(15)70044-7.
20. D‘Ancona F, Caporali MG, Del Manso M, et al. Invasive pneumococcal disease in children and adults in seven Italian regions after the introduction of the conjugate vaccine, 2008–2014. Epidemiol Prev, 2015;39(4):134–138.
21. Moore MR, Link-Gelles R, Schaffner W, et al. Effect of use of 13-valent pneumococcal conjugate vaccine in children on invasive pneumococcal disease in children and adults in the USA: analysis of multisite, population-based surveillance. Lancet Infect Dis., 2015;15(3):301–309. doi: 10.1016/S1473-3099(14)71081-3. Epub 2015 Feb 3. PMID: 25656600; PMCID: PMC4876855.
22. Savulescu C, Krizova P, Lepoutre A, et al. Effect of high-valency pneumococcal conjugate vaccines on invasive pneumococcal disease in children in SpIDnet countries: an observational multicentre study. Lancet Respir Med., 2017;5(8):648–656. doi: 10.1016/ S2213-2600(17)30110-8. Epub 2017 Mar 27. PMID: 28359798.
23. Invasive Pneumococcal Disease in the Czech Republic. Reports of the Centre for Epidemiology and Microbiology. Available at: https://szu.gov.cz/temata-zdravi-a - bezpecnosti/a-z-infekce/s/ streptokokova-infekce/invazivni-pneumokokova-onemocneni-v-ceske-republice/ (Access: 22.7.2025).
24. Kozáková J, Křížová P, Malý M. Impact of pneumococcal conjugate vaccine on invasive pneumococcal disease in children under 5 years of age in the Czech Republic. PLoS One, 2021;16(2):e0247862. DOI: 10.1371/journal.pone.0247862.
25. Weinberger DM, Malley R, Lipsitch M. Serotype replacement in disease after pneumococcal vaccination. Lancet, 2011;378(9807):1962–1973. doi: 10.1016/S0140-6736(10)62225 - 8. Epub 2011 Apr 12. PMID: 21492929; PMCID: PMC3256741.
26. Principi N, Di Cara G, Bizzarri I, et al. Prevention of invasive pneumococcal disease: Problems emerged after some years of the 13-valent pneumococcal conjugate vaccine use. Curr Infect Dis Rep, 2018;20(1):1. DOI: 10.1007/s11908-018-0607-z.
27. Brueggemann AB, Jansen van Rensburg MJ, Shaw D, et al. Changes in the incidence of invasive disease due to Streptococcus pneumoniae, Haemophilus influenzae, and Neisseria meningitidis during the COVID-19 pandemic in 26 countries and territories in the Invasive Respiratory Infection Surveillance Initiative: a prospective analysis of surveillance data. Lancet Digit Health., 2021;3(6):e360–e370. doi: 10.1016/S2589 - 7500(21)00077-7. Erratum in: Lancet Digit Health. 2021 Jul;3(7):e413. doi: 10.1016/ S2589-7500(21)00103-5. PMID: 34045002; PMCID: PMC8166576.
28. Shaw D, Abad R, Amin-Chowdhury Z, et al. Trends in invasive bacterial diseases during the first 2 years of the COVID-19 pandemic: analyses of prospective surveillance data from 30 countries and territories in the IRIS Consortium. Lancet Digit Health., 2023;5(9):e582–e593. doi: 10.1016/S2589-7500(23)00108-5. Epub 2023 Jul 27. PMID: 37516557; PMCID: PMC10914672.
29. WHO Immunization coverage. Available at: https://www.who. int/news-room/fact - sheets/detail/immunization-coverage (Access: 22.7.2025).
30. Cohen R, Ashman M, Taha MK, et al. Pediatric Infectious Disease Group (GPIP) position paper on the immune debt of the COVID-19 pandemic in childhood, how can we fill the immunity gap? Infect Dis Now., 2021;51(5):418–423. doi: 10.1016/j.idnow.2021.05.004. Epub 2021 May 12. PMID: 33991720; PMCID: PMC8114587.
31. McQuaid F, Mulholland R, Sangpang Rai Y, et al. Uptake of infant and preschool immunisations in Scotland and England during the COVID-19 pandemic: An observational study of routinely collected data. PLoS Med., 2022;19(2):e1003916. doi: 10.1371/ journal.pmed.1003916. PMID: 35192611; PMCID: PMC8863286.
32. Andrews NJ, Waight PA, Burbidge P, et al. Serotype-specific effectiveness and correlates of protection for the 13-valent pneumococcal conjugate vaccine: a postlicensure indirect cohort study. Lancet Infect Dis., 2014;14(9):839–846. doi: 10.1016/ S1473-3099(14)70822-9. Epub 2014 Jul 17. PMID: 25042756.
33. Slotved HC, Dalby T, Harboe ZB, Valentiner-Branth P, Casadevante VF, Espenhain L, Fuursted K, Konradsen HB. The incidence of invasive pneumococcal serotype 3 disease in the Danish population is not reduced by PCV-13 vaccination. Heliyon, 2016;2(11):e00198. doi: 10.1016/j.heliyon.2016.e00198. PMID: 27957553; PMCID: PMC5133732.
34. Wijayasri S, Hillier K, Lim GH, et al. The shifting epidemiology and serotype distribution of invasive pneumococcal disease in Ontario, Canada, 2007–2017. PLoS One, 2019;14(12):e0226353. doi: 10.1371/journal.pone.0226353. PMID: 31834926; PMCID: PMC6910703.
35. Ladhani SN, Collins S, Djennad A, et al. Rapid increase in non-vaccine serotypes causing invasive pneumococcal disease in England and Wales, 2000–2017: a prospective national observational cohort study. Lancet Infect Dis., 2018;18(4):441–451. doi: 10.1016/S1473-3099(18)30052-5. Epub 2018 Jan 26. Erratum in: Lancet Infect Dis. 2018 Apr;18(4):376. doi: 10.1016/ S1473-3099(18)30074-4. PMID: 29395999.
36. Andrews N, Kent A, Amin-Chowdhury Z, et al. Effectiveness of the seven-valent and thirteen-valent pneumococcal conjugate vaccines in England: The indirect cohort design, 2006 – 2018. Vaccine, 2019;37(32):4491–4498. DOI: 10.1016/j.vaccine.2019.06.071.
37. Lupinacci R, Rupp R, Wittawatmongkol O, et al. A phase 3, multicenter, randomized, double-blind, active-comparator-controlled study to evaluate the safety, tolerability, and immunogenicity of a 4-dose regimen of V114, a 15-valent pneumococcal conjugate vaccine, in healthy infants (PNEUPED). Vaccine, 2023;41(5):1142–1152. doi: 10.1016/j.vaccine.2022.12.054. Epub 2023 Jan 6. PMID: 36621410.
38. Wagner G, Gartlehner G, Thaler K, et al. Immunogenicity and safety of the 15-valent pneumococcal conjugate vaccine, a systematic review and meta-analysis. Vaccines, 2024; 9 : 257. Avalable at: https://doi.org/10.1038/s41541-024-01048-y.
39. Kellner JD, Ricketson LJ, Demczuk WHB, et al. Whole-Genome Analysis of Streptococcus pneumoniae Serotype 4 Causing Outbreak of Invasive Pneumococcal Disease, Alberta, Canada. Emerg Infect Dis., 2021;27(7):1867–1875. doi: 10.3201/eid2707.204403. PMID: 34152965; PMCID: PMC8237880.
40. Gladstone RA, Siira L, Brynildsrud OB, et al. International links between Streptococcus pneumoniae vaccine serotype 4 sequence type (ST) 801 in Northern European shipyard outbreaks of invasive pneumococcal disease. Vaccine, 2022;40(7):1054 – 1060. doi: 10.1016/j.vaccine.2021.10.046. Epub 2022 Jan 5. PMID: 34996643; PMCID: PMC8820377.
41. Kobayashi M, Leidner AJ, Gierke R, et al. Use of 21-Valent Pneumococcal Conjugate Vaccine Among U.S. Adults: Recommendations of the Advisory Committee on Immunization Practices – United States, 2024. MMWR Morb Mortal Wkly Rep., 2024;73(36):793–798. doi: 10.15585/mmwr.mm7336a3. PMID: 39264843; PMCID: PMC11392227.
42. Schellenberg JJ, Adam HJ, Baxter MR, et al. Comparing serotype coverage of pneumococcal vaccines with PCV21 (V116), a new 21-valent conjugate pneumococcal vaccine, and the epidemiology of its eight unique Streptococcus pneumoniae serotypes (15A, 15C, 16F, 23A, 23B, 24F, 31 and 35B) causing invasive pneumococcal disease in adult patients in Canada: SAVE study, 2018–21. J Antimicrob Chemother., 2025;80(5):1377–1385. doi: 10.1093/jac/dkaf085. PMID: 40131289; PMCID: PMC12046396.
43. Antimicrobial resistance in the EU/EEA (EARS-Net) – Annual Epidemiological Report 2023. Available at: https://www.ecdc. europa.eu/sites/default/files/documents/antimicrobial-resistance - annual-epidemiological-report-EARS-Net-2023.pdf (Access: 22.7.2025).
44. Invasive pneumococcal disease – Annual Epidemiological Report for 2022. Available at: https://www.ecdc.europa.eu/sites/ default/files/documents/PNEU_AER_2022_Report.p df (Access: 1.10.2025)
45. Vaccine schedules in all countries in the EU/EEA. Available at: https://vaccine - schedule.ecdc.europa.eu/ (Access: 1.10.2025).
Labels
Hygiene and epidemiology Medical virology Clinical microbiologyArticle was published in
Epidemiology, Microbiology, Immunology

-
All articles in this issue
- Epidemiology and spatial analysis of hepatitis B and C in the Czech Republic
- Parvovirus B19: how to interpret semi-quantitative viraemia values during high circulation of the virus in the population
- Surveillance of invasive pneumococcal disease and serotypes coverage of pneumococcal vaccines in the Czech Republic between 2007 and 2024
- Usutu virus meningitis: another overlooked mosquito-borne disease in central Europe
- Epidemiology, Microbiology, Immunology
- Journal archive
- Current issue
- About the journal
Most read in this issue
- Surveillance of invasive pneumococcal disease and serotypes coverage of pneumococcal vaccines in the Czech Republic between 2007 and 2024
- Usutu virus meningitis: another overlooked mosquito-borne disease in central Europe
- Epidemiology and spatial analysis of hepatitis B and C in the Czech Republic
- Parvovirus B19: how to interpret semi-quantitative viraemia values during high circulation of the virus in the population
